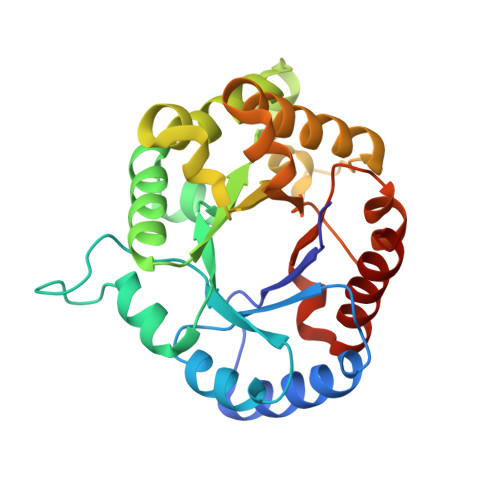
Structure of triosephosphate isomerase from Escherichia coli determined at 2.6 A resolution.
Noble, M.E., Zeelen, J.P., Wierenga, R.K., Mainfroid, V., Goraj, K., Gohimont, A.C., Martial, J.A.(1993) Acta Crystallogr D Biol Crystallogr 49: 403-417
- PubMed: 15299515 Search on PubMed
- DOI: https://doi.org/10.1107/S0907444993002628
- Primary Citation Related Structures:
1TRE - PubMed Abstract:
The structure of triosephosphate isomerase (TIM) from the organism Escherichia coli has been determined at a resolution of 2.6 A. The structure was solved by the molecular replacement method, first at 2.8 A resolution with a crystal grown by the technique of hanging-drop crystallization from a mother liquor containing the transition-state analogue 2-phosphoglycolate (2PG). As a search model in the molecular replacement calculations, the refined structure of TIM from Trypanosoma brucei, which has a sequence identity of 46% compared to the enzyme from E. coli, was used. An E. coli TIM crystal grown in the absence of 2PG, diffracting to 2.6 A resolution, was later obtained by application of the technique of macro-seeding using a seed crystal grown from a mother liquor without 2PG. The final 2.6 A model has a crystallographic R factor of 11.9%, and agrees well with standard stereochemical parameters. The structure of E. coli TIM suggests the importance of residues which favour helix initiation for the formation of the TIM fold. In addition, TIM from E. coli shows peculiarities in its dimer interface, and in the packing of core residues within the beta-barrel.
- European Molecular Biology Laboratory, Heidelburg, Germany.
Organizational Affiliation: