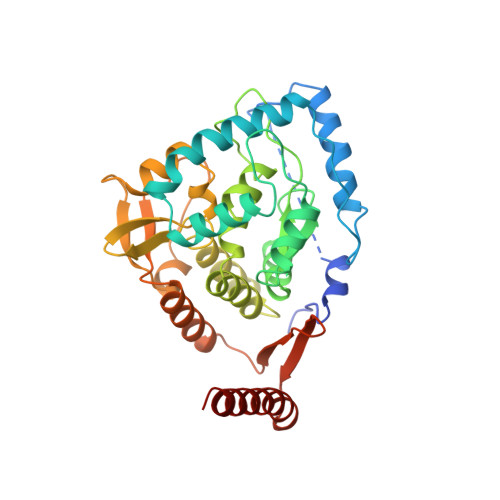
Crystal structure of tyrosine hydroxylase at 2.3 A and its implications for inherited neurodegenerative diseases.
Goodwill, K.E., Sabatier, C., Marks, C., Raag, R., Fitzpatrick, P.F., Stevens, R.C.(1997) Nat Struct Biol 4: 578-585
- PubMed: 9228951 Search on PubMed
- DOI: https://doi.org/10.1038/nsb0797-578
- Primary Citation Related Structures:
1TOH - PubMed Abstract:
Tyrosine hydroxylase (TyrOH) catalyzes the conversion of tyrosine to L-DOPA, the rate-limiting step in the biosynthesis of the catecholamines dopamine, adrenaline, and noradrenaline. TyrOH is highly homologous in terms of both protein sequence and catalytic mechanism to phenylalanine hydroxylase (PheOH) and tryptophan hydroxylase (TrpOH). The crystal structure of the catalytic and tetramerization domains of TyrOH reveals a novel alpha-helical basket holding the catalytic iron and a 40 A long anti-parallel coiled coil which forms the core of the tetramer. The catalytic iron is located 10 A below the enzyme surface in a 17 A deep active site pocket and is coordinated by the conserved residues His 331, His 336 and Glu 376. The structure provides a rationale for the effect of point mutations in TyrOH that cause L-DOPA responsive parkinsonism and Segawa's syndrome. The location of 112 different point mutations in PheOH that lead to phenylketonuria (PKU) are predicted based on the TyrOH structure.
- Department of Chemistry, University of California, Berkeley 94720, USA.
Organizational Affiliation: