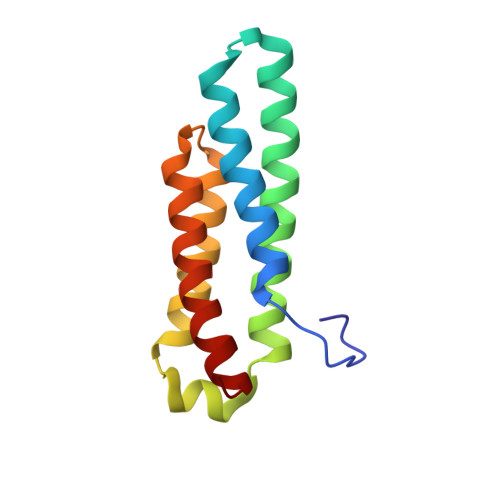
Nickel superoxide dismutase structure and mechanism.
Barondeau, D.P., Kassmann, C.J., Bruns, C.K., Tainer, J.A., Getzoff, E.D.(2004) Biochemistry 43: 8038-8047
- PubMed: 15209499 Search on PubMed
- DOI: https://doi.org/10.1021/bi0496081
- Primary Citation Related Structures:
1T6I, 1T6Q, 1T6U - PubMed Abstract:
The 1.30 A resolution crystal structure of nickel superoxide dismutase (NiSOD) identifies a novel SOD fold, assembly, and Ni active site. NiSOD is a hexameric assembly of right-handed 4-helix bundles of up-down-up-down topology with N-terminal hooks chelating the active site Ni ions. This newly identified nine-residue Ni-hook structural motif (His-Cys-X-X-Pro-Cys-Gly-X-Tyr) provides almost all interactions critical for metal binding and catalysis, and thus will likely be diagnostic of NiSODs. Conserved lysine residues are positioned for electrostatic guidance of the superoxide anion to the narrow active site channel. Apo structures show that the Ni-hook motif is unfolded prior to metal binding. The active site Ni geometry cycles from square planar Ni(II), with thiolate (Cys2 and Cys6) and backbone nitrogen (His1 and Cys2) ligands, to square pyramidal Ni(III) with an added axial His1 side chain ligand, consistent with electron paramagentic resonance spectroscopy. Analyses of the three NiSOD structures and comparisons to the Cu,Zn and Mn/Fe SODs support specific molecular mechanisms for NiSOD maturation and catalysis, and identify important structure-function relationships conserved among SODs.
- Department of Molecular Biology, The Skaggs Institute for Chemical Biology, The Scripps Research Institute, 10550 North Torrey Pines Road, La Jolla, California 92037, USA.
Organizational Affiliation: