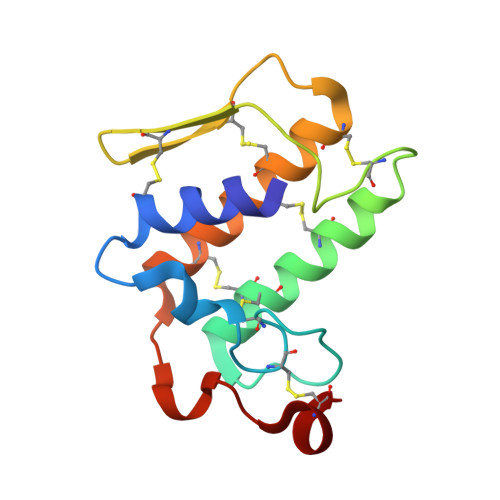
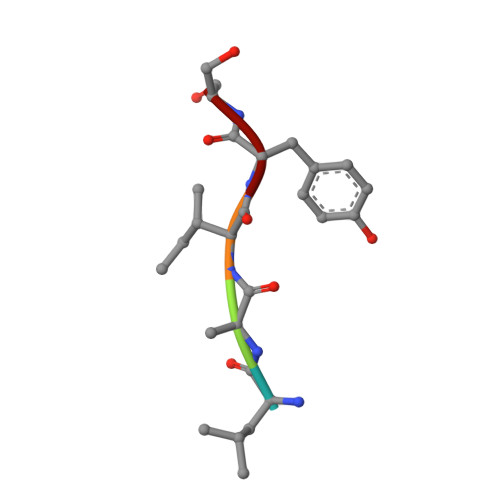
Crystal structure of the complex of group I PLA2 with a group II-specific peptide Leu-Ala-Ile-Tyr-Ser (LAIYS) at 2.6 A resolution.
Singh, R.K., Singh, N., Jabeen, T., Sharma, S., Dey, S., Singh, T.P.(2005) J Drug Target 13: 367-374
- PubMed: 16278156
- DOI: https://doi.org/10.1080/10611860500254450
- Primary Citation of Related Structures:
1T37, 1ZM6 - PubMed Abstract:
Phospholipases A(2)s (PLA(2)s) are widely distributed in mammals and snake venoms. They catalyze the production of arachidonic acid from membrane phospholipids leading to the bioynthesis of pro-inflammatory eicosanoids. A peptide Leu-Ala-Ile-Tyr-Ser (LAIYS) was designed and synthesized as a specific inhibitor of PLA(2). It was shown earlier that the peptide bound to group II PLA(2) specifically and had a dissociation constant (K(d)) of 8.8 x 10(-9) M. In the present studies for the binding of LAIYS with a group I PLA(2) from Naja naja sagittifera using surface plasmon resonance the dissociation constant was found to be 4.5 x 10(-5) M which is considerably lower than the value found for the group II PLA(2). In order to determine the details of binding at the molecular level, a group I PLA(2) from the venom of Naja naja sagittifera was crystallized with peptide LAIYS. The crystal structure showed the presence of LAIYS at the substrate-binding site but has fewer interactions than those observed with group II PLA(2) from Daboia russelli pulchella. The observed difference in the binding affinity is caused primarily due to poor fitting of the peptide LAIYS in the binding site of group I PLA(2). Apparently, the location of Trp 19 in group I PLA(2) is not favourable for the binding of LAIYS. The two complexes also differ drastically in the formation of intermolecular interactions. In the present structure, the side chain of Ser (P) interacts with His 48 and Asp 49 while in the complex with group II PLA(2) it was Tyr (P) OH that formed the corresponding interactions. Tyr (P) in group I PLA(2) is the main contributor of the hydrophobic interactions whereas in the complex of LAIYS with group II PLA(2) it was the peptide segment Leu-Ala-Ile that produced the bulk of hydrophobic forces. The structures further showed that the peptide LAIYS was fully inside the substrate-binding region of the group II PLA(2) while a significant portion of the peptide LAIYS was hanging outside the surface of the group I PLA(2). The buried area in the complex with group II PLA(2) was 811 A(2) whereas, the corresponding area in group I PLA(2) was 449 A(2). This shows that the peptide LAIYS is very compatible with the substrate-binding site of group II PLA(2) and rather poorly fits into the substrate-binding site of group I PLA(2). This indicates that a highly specific ligand for one form of PLA(2) may be a poor partner for another form of enzyme.
- All India Institute of Medical Sciences, Department of Biophysics, New Delhi, India.
Organizational Affiliation: