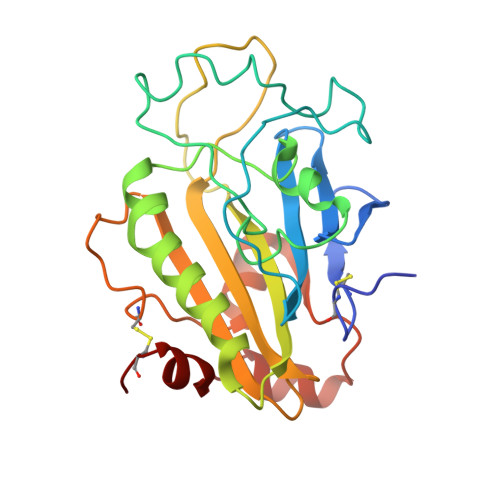
Identification of the Serratia endonuclease dimer: structural basis and implications for catalysis.
Miller, M.D., Krause, K.L.(1996) Protein Sci 5: 24-33
- PubMed: 8771193 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1002/pro.5560050104
- Primary Citation Related Structures:
1SMN - PubMed Abstract:
The Serratia endonuclease is an extracellularly secreted enzyme capable of cleaving both single- and double-stranded forms of DNA and RNA. It is the first member of a large class of related and usually dimeric endonucleases for which a structure is known. Using X-ray crystallography, the structure of monomer of this enzyme was reported by us previously (Miller MD et al., 1994, Nature Struct Biol 1:461-468). We now confirm the dimeric nature of this enzyme through light-scattering experiments and identify the physiologic dimer interface through crystal packing analysis. This dimerization occurs through an isologous twofold interaction localized to the carboxy-terminal subdomain of the enzyme. The dimer is a prolate ellipsoid with dimensions 30 A x 35 A x 90 A. The dimer interface is flat and contains four salt links, several hydrogen bonds, and nonpolar interactions. Buried water is prominent in this interface and it includes an unusual "cubic" water cluster. The position of the two active sites in the dimer suggests that they can act independently in their cleavage of DNA, but have a geometrical advantage in attacking substrate relative to the monomer.
- Department of Biochemical and Biophysical Sciences, University of Houston, Texas 77204-5934, USA.
Organizational Affiliation: