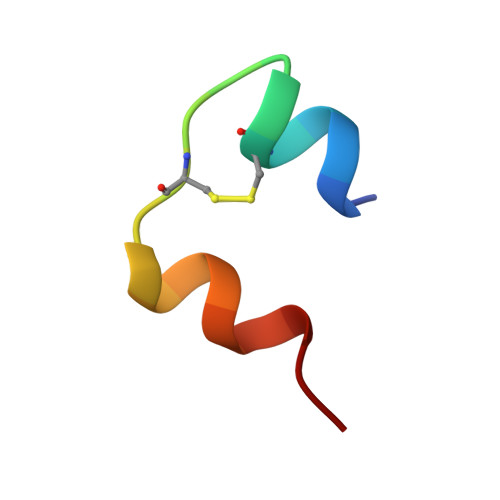
Structure of monomeric porcine DesB1-B2 despentapeptide (B26-B30) insulin at 1.65 A resolution.
Diao, J.S., Wan, Z.L., Chang, W.R., Liang, D.C.(1997) Acta Crystallogr D Biol Crystallogr 53: 507-512
- PubMed: 15299880 Search on PubMed
- DOI: https://doi.org/10.1107/S0907444997004034
- Primary Citation Related Structures:
1SDB - PubMed Abstract:
Insulin has a concentration of 10(-8)-10(-11) M in the blood which ensures that it circulates and exerts its physiological functions in vivo as a monomer. The crystal structure of monomeric porcine desB1-B2 despentapeptide (B26-B30) insulin (DesB1-2 DPI) with M(r) = 4934 Da has been determined at 1.65 A resolution using the molecular replacement method. A structural comparison between DesB1-2 DPI and 2Zn insulin reveals that the conformation of DesB1-2 DPI is more similar to molecule I than molecule II of 2Zn insulin. The remarkable conformational difference between B25-Phe in DesB1-2 DPI and B25-Phe in despentapeptide (B26-B30) insulin (DPI) indicates that the residue B25-Phe possesses great flexibility and mobility.
- National Laboratory of Biomacromolecules, Institute of Biophysics, Chinese Academy of Sciences, Beijing, China.
Organizational Affiliation: