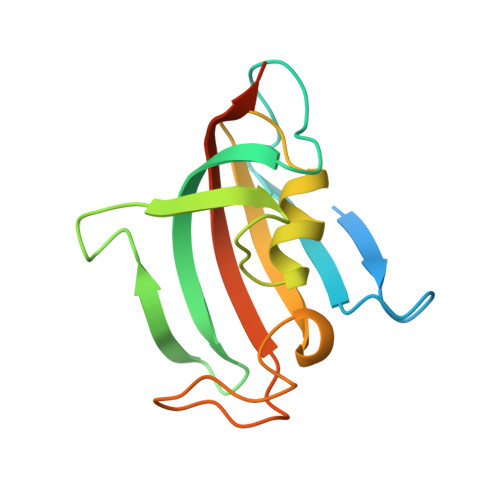
Three-dimensional structure of the immunophilin-like domain of FKBP59 in solution.
Craescu, C.T., Rouviere, N., Popescu, A., Cerpolini, E., Lebeau, M.C., Baulieu, E.E., Mispelter, J.(1996) Biochemistry 35: 11045-11052
- PubMed: 8780506 Search on PubMed
- DOI: https://doi.org/10.1021/bi960975p
- Primary Citation Related Structures:
1ROT, 1ROU - PubMed Abstract:
FKBP59 is a protein usually associated with heat-shock protein hsp90 and steroid receptors. The N-terminal domain of the rabbit liver protein (149 amino acids) has a sequence homology with FKBP12, binds FK506 immunosuppressor, and has a peptidyl-prolyl cis-trans isomerase activity. The three-dimensional structure of this domain (FKBP59-I) was determined using homo- and heteronuclear multidimensional NMR spectroscopy, distance geometry, and molecular dynamics methods. Structure calculations used 1290 interproton distance restraints derived from nuclear Overhauser enhancement measurements, 29 dihedral phi angle restraints, and 92 hydrogen bond restraints. For the final 22 structures, the root mean square distance from the mean atomic coordinates, calculated for well-defined secondary structure fragments, is 0.47 +/- 0.05 and 1.26 +/- 0.15 A for backbone heavy atoms (N, C alpha, C') and for all non-hydrogen atoms, respectively. The global fold contains a twisted six-stranded antiparallel beta-sheet and a short alpha-helix packed on the hydrophobic side of the sheet. The 20 N-terminal and 12 C-terminal amino acids of the domain are disordered. The main-chain structure of FKBP59-I is globally similar to the NMR-derived and X-ray structures of unbound FKBP12. An unusual hydrogen bond interaction between the indole amino proton of Trp 89 and the aromatic cycle of Phe 129 was observed. This gives a large upfield shift (-4.8 ppm) and a significant exchange protection factor. The implications of the present structure determination on the ligand binding of FKBP59 are discussed.
- Institut National de la Santé et de la Recherche Médicale U350, Institut Curie, Orsay, France. craescu@curie.u-psud.fr
Organizational Affiliation: