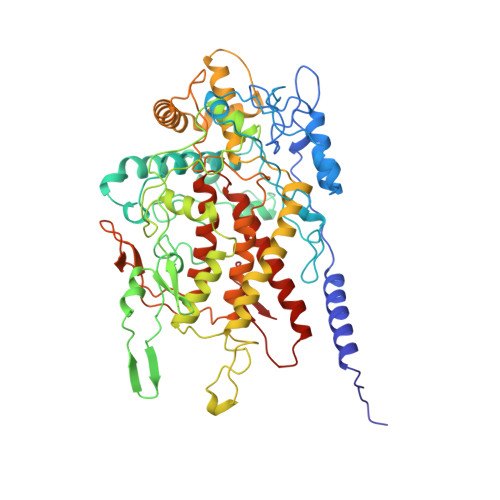
Crystal structure of dodecameric vanadium-dependent bromoperoxidase from the red algae Corallina officinalis.
Isupov, M.N., Dalby, A.R., Brindley, A.A., Izumi, Y., Tanabe, T., Murshudov, G.N., Littlechild, J.A.(2000) J Mol Biology 299: 1035-1049
- PubMed: 10843856 Search on PubMed
- DOI: https://doi.org/10.1006/jmbi.2000.3806
- Primary Citation Related Structures:
1QHB - PubMed Abstract:
The three-dimensional structure of the vanadium bromoperoxidase protein from the marine red macroalgae Corallina officinalis has been determined by single isomorphous replacement at 2.3 A resolution. The enzyme subunit is made up of 595 amino acid residues folded into a single alpha+beta domain. There are 12 bromoperoxidase subunits, arranged with 23-point group symmetry. A cavity is formed by the N terminus of each subunit in the centre of the dodecamer. The subunit fold and dimer organisation of the Cor. officinalis vanadium bromoperoxidase are similar to those of the dimeric enzyme from the brown algae Ascophyllum nodosum, with which it shares 33 % sequence identity. The different oligomeric state of the two algal enzymes seems to reflect separate mechanisms of adaptation to harsh environmental conditions and/or to chemically active substrates and products. The residues involved in the vanadate binding are conserved between the two algal bromoperoxidases and the vanadium chloroperoxidase from the fungus Curvularia inaequalis. However, most of the other residues forming the active-site cavity are different in the three enzymes, which reflects differences in the substrate specificity and stereoselectivity of the reaction. A dimer of the Cor. officinalis enzyme partially superimposes with the two-domain monomer of the fungal enzyme.
- Schools of Chemistry and Biological Sciences, University of Exeter, Stocker Road, Exeter, EX4 4QD, UK.
Organizational Affiliation: