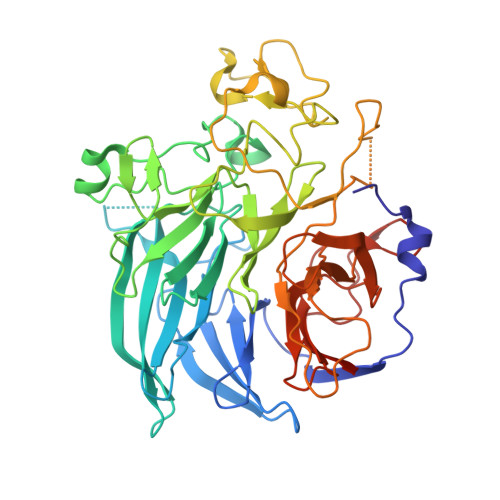
The 1.7 A crystal structure of the apo form of the soluble quinoprotein glucose dehydrogenase from Acinetobacter calcoaceticus reveals a novel internal conserved sequence repeat.
Oubrie, A., Rozeboom, H.J., Kalk, K.H., Duine, J.A., Dijkstra, B.W.(1999) J Mol Biology 289: 319-333
- PubMed: 10366508 Search on PubMed
- DOI: https://doi.org/10.1006/jmbi.1999.2766
- Primary Citation Related Structures:
1QBI - PubMed Abstract:
The crystal structure of a dimeric apo form of the soluble quinoprotein glucose dehydrogenase (s-GDH) from Acinetobacter calcoaceticus has been solved by multiple isomorphous replacement followed by density modification, and was subsequently refined at 1. 72 A resolution to a final crystallographic R-factor of 16.5% and free R-factor of 20.8% [corrected]. The s-GDH monomer has a beta-propeller fold consisting of six four-stranded anti-parallel beta-sheets aligned around a pseudo 6-fold symmetry axis. The enzyme binds three calcium ions per monomer, two of which are located in the dimer interface. The third is bound in the putative active site, where it may bind and functionalize the pyrroloquinoline quinone (PQQ) cofactor. A data base search unexpectedly showed that four uncharacterized protein sequences are homologous to s-GDH with many residues in the putative active site absolutely conserved. This indicates that these homologs may have a similar structure and that they may catalyze similar PQQ-dependent reactions.A structure-based sequence alignment of the six four-stranded beta-sheets in s-GDH's beta-propeller fold shows an internally conserved sequence repeat that gives rise to two distinct conserved structural motifs. The first structural motif is found at the corner of the short beta-turn between the inner two beta-strands of the beta-sheets, where an Asp side-chain points back into the beta-sheet to form a hydrogen-bond with the OH/NH of a Tyr/Trp side-chain in the same beta-sheet. The second motif involves an Arg/Lys side-chain in the C beta-strand of one beta-sheet, which forms a bidentate salt-bridge with an Asp/Glu in the CD loop of the next beta-sheet. These intra and inter-beta-sheet hydrogen-bonds are likely to contribute to the stability of the s-GDH beta-propeller fold.
- Laboratory of Biophysical Chemistry and BIOSON Research Institute, University of Groningen, Nijenborgh 4, Groningen, 9747 AG, The Netherlands.
Organizational Affiliation: