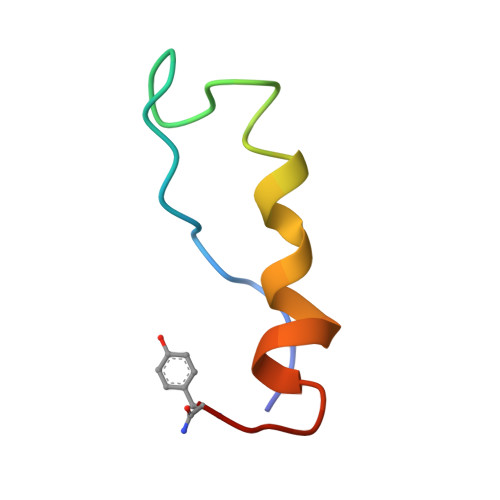
Solution structure of monomeric peptide YY supports the functional significance of the PP-fold.
Keire, D.A., Kobayashi, M., Solomon, T.E., Reeve, J.R.(2000) Biochemistry 39: 9935-9942
- PubMed: 10933813 Search on PubMed
- DOI: https://doi.org/10.1021/bi992576a
- Primary Citation Related Structures:
1QBF - PubMed Abstract:
Peptide YY (PYY) belongs to a family of peptides including neuropeptide Y (NPY) and pancreatic peptide (PP) that regulate numerous functions through both central and peripheral receptors. The solution structure of these peptides is hypothesized to be critically important in receptor selectivity and activation, based on prior demonstration of a stable tertiary conformation of PP called the "PP-fold". Circular dichroism (CD) spectra show a pH-dependent structural transition in the pH range 3-4. Thus we describe the tertiary structure of porcine PYY in water at pH 5.5, 25 degrees C, and 150 mM NaCl, as determined from 2D (1)H NMR data recorded at 500 MHz. A constraint set consisting of 396 interproton distances from NOE data was used as input for distance geometry, simulated annealing, and restrained energy minimization calculations in X-PLOR. The RMSDs of the 20 X-PLOR-generated structures were 0.71 +/- 0.14 and 1.16 +/- 0.17 A, respectively, for backbone and heavy atom overlays of residues 1-34. The resulting structure consists of two C-terminal helical segments from residues 17 to 22 and 25 to 33 separated by a kink at residues 23, 24, and 25, a turn centered around residues 12-14, and the N-terminus folded near residues 30 and 31. The well-defined portions of the PYY structure reported here bear a marked similarity to the structure of PP. Our findings strongly support the importance of the stable folded structure of this family of peptides for binding and activation of Y receptor subtypes.
- CURE Digestive Diseases Research Center, Greater Los Angeles Veterans Healthcare System, Los Angeles, California 90073, USA. dkeire@ucla.edu
Organizational Affiliation: