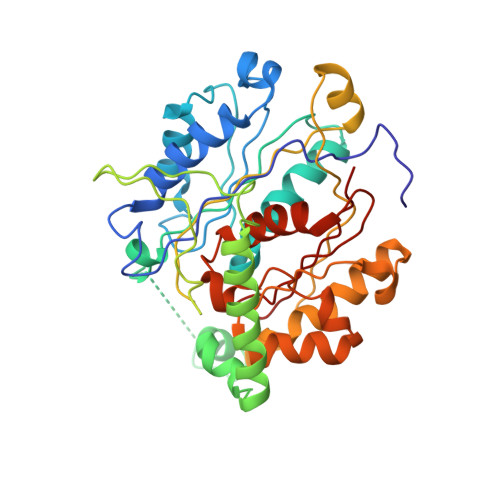
The 2.8 A crystal structure of peroxisomal 3-ketoacyl-CoA thiolase of Saccharomyces cerevisiae: a five-layered alpha beta alpha beta alpha structure constructed from two core domains of identical topology.
Mathieu, M., Zeelen, J.P., Pauptit, R.A., Erdmann, R., Kunau, W.H., Wierenga, R.K.(1994) Structure 2: 797-808
- PubMed: 7812714 Search on PubMed
- DOI: https://doi.org/10.1016/s0969-2126(94)00081-6
- Primary Citation Related Structures:
1PXT - PubMed Abstract:
The peroxisomal enzyme 3-ketoacyl-coenzyme A thiolase of the yeast Saccharomyces cerevisiae is a homodimer with 417 residues per subunit. It is synthesized in the cytosol and subsequently imported into the peroxisome where it catalyzes the last step of the beta-oxidation pathway. We have determined the structure of this thiolase in order to study the reaction mechanism, quaternary associations and intracellular targeting of thiolases generally, and to understand the structural basis of genetic disorders associated with human thiolases. Here we report the crystal structure of unliganded yeast thiolase refined at 2.8 A resolution. The enzyme comprises three domains; two compact core domains having the same fold and a loop domain. Each of the two core domains is folded into a mixed five-stranded beta-sheet covered on each side by helices and the two are assembled into a five-layered alpha beta alpha beta alpha structure. The central layer is formed by two helices, which point with their amino termini towards the active site. The loop domain, which is to some extent stabilized by interactions with the other subunit, runs over the surface of the two core domains, encircling the active site of its own subunit. The crystal structure of thiolase shows that the active site is a shallow pocket, shaped by highly conserved residues. Two conserved cysteines and a histidine at the floor of this pocket probably play key roles in the reaction mechanism. The two active sites are on the same face of the dimer, far from the amino and carboxyl termini of both subunits and the disordered amino-terminal import signal sequence.
- EMBL, Heidelberg, Germany.
Organizational Affiliation: