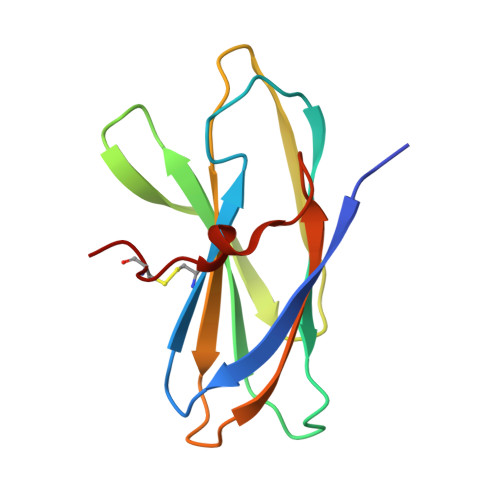
Crystal and Solution Structures of a Superantigen from Yersinia pseudotuberculosis Reveal a Jelly-Roll Fold.
Donadini, R., Liew, C.W., Kwan, A.H., Mackay, J.P., Fields, B.A.(2004) Structure 12: 145-156
- PubMed: 14725774 Search on PubMed
- DOI: https://doi.org/10.1016/j.str.2003.12.002
- Primary Citation Related Structures:
1PM4, 1POQ - PubMed Abstract:
Superantigens are a class of microbial proteins with the ability to excessively activate T cells by binding to the T cell receptor. The staphylococcal and streptococcal superantigens are closely related in structure and possess an N-terminal domain that resembles an OB fold and a C-terminal domain similar to a beta-grasp fold. Yersinia pseudotuberculosis produces superantigens, YPMa, YPMb, and YPMc, which have no significant amino acid similarity to other proteins. We have determined the crystal and solution structures of YPMa, which show that the protein has a jelly-roll fold. The closest structural neighbors to YPMa are viral capsid proteins and members of the tumor necrosis factor superfamily. In the crystal structure, YPMa packs as a trimer, another feature shared with viral capsid proteins and TNF superfamily proteins. However, in solution YPMa behaves as a monomer, and any functional relevance of the trimer observed in the crystals is yet to be established.
- School of Molecular and Microbial Biosciences, University of Sydney, Sydney, New South Wales 2006, Australia.
Organizational Affiliation: