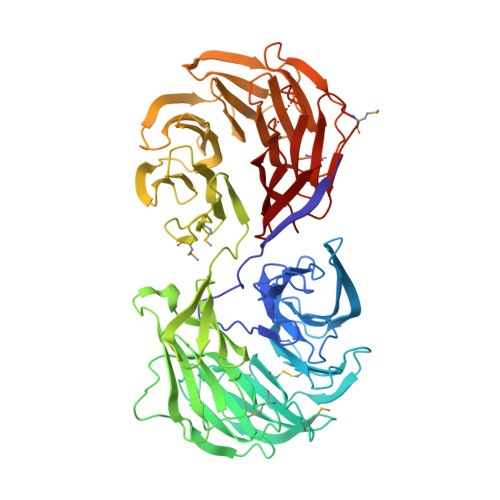
The structure of Aip1p, a WD repeat protein that regulates Cofilin-mediated actin depolymerization.
Voegtli, W.C., Madrona, A.Y., Wilson, D.K.(2003) J Biological Chem 278: 34373-34379
- PubMed: 12807914 Search on PubMed
- DOI: https://doi.org/10.1074/jbc.M302773200
- Primary Citation Related Structures:
1PGU, 1PI6 - PubMed Abstract:
Actin-interacting protein 1 (Aip1p) is a 67-kDa WD repeat protein known to regulate the depolymerization of actin filaments by cofilin and is conserved in organisms ranging from yeast to mammals. The crystal structure of Aip1p from Saccharomyces cerevisiae was determined to a 2.3-A resolution and a final crystallographic R-factor of 0.204. The structure reveals that the overall fold is formed by two connected seven-bladed beta-propellers and has important implications for the structure of Aip1 from other organisms and WD repeat-containing proteins in general. These results were unexpected because a maximum of 10 WD repeats had been reported in the literature for this protein using sequence data. The surfaces of the beta-propellers formed by the D-A and B-C loops are positioned adjacent to one another, giving Aip1p a shape that resembles an open "clamshell." The mapping of conserved residues to the structure of Aip1p reveals dense patches of conserved residues on the surface of one beta-propeller and at the interface of the two beta-propellers. These two patches of conserved residues suggest a potential binding site for F-actin on Aip1p and that the orientation of the beta-propellers with respect to one another plays a role in binding an actin-cofilin complex. In addition, the conserved interface between the domains is mediated by a number of interactions that appear to impart rigidity between the two domains of Aip1p and may make a large substrate-induced conformational change difficult.
- Section of Molecular and Cellular Biology, University of California, Davis, California 95616, USA.
Organizational Affiliation: