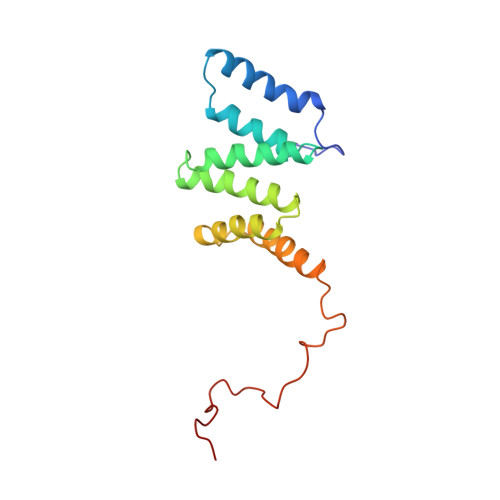
The Solution Structure of Human Mitochondria Fission Protein Fis1 Reveals a Novel TPR-like Helix Bundle
Suzuki, M., Jeong, S.Y., Karbowski, M., Youle, R.J., Tjandra, N.(2003) J Mol Biology 334: 445-458
- PubMed: 14623186 Search on PubMed
- DOI: https://doi.org/10.1016/j.jmb.2003.09.064
- Primary Citation Related Structures:
1PC2 - PubMed Abstract:
Fis1 in yeast localizes to the outer mitochondrial membrane and facilitates mitochondrial fission by forming protein complexes with Dnm1 and Mdv1. Fis1 orthologs exist in higher eukaryotes, suggesting that they are functionally conserved. In the present study, we cloned the human Fis1 ortholog that was predicted in a database, and determined the protein structure using NMR spectroscopy. Following a flexible N-terminal tail, six alpha-helices connected with short loops construct a single core domain. The C-terminal tail containing a transmembrane segment appears to be disordered. In the core domain, each of two sequentially adjacent helices forms a hairpin-like conformation, resulting in a six helix assembly forming a slightly twisted slab similar to that of a tandem array of tetratrico-peptide repeat (TPR) motif folds. Within this TPR-like core domain, no significant sequence similarity to the typical TPR motif is found. The structural analogy to the TPR-containing proteins suggests that Fis1 binds to other proteins at its concave hydrophobic surface. A simple composition of Fis1 comprised of a binding domain and a transmembrane segment indicates that the protein may function as a molecular adaptor on the mitochondrial outer membrane. In HeLa cells, however, increased levels in mitochondria-associated Fis1 did not result in mitochondrial translocation of Drp1, a potential binding partner of Fis1 implicated in the regulation of mitochondrial fission, suggesting that the interaction between Drp1 and Fis1 is regulated.
- Biochemistry Section, Surgical Neurology Branch, National Institute of Neurological Disorders and Stroke, National Institutes of Health, Bethesda, MD 20892, USA.
Organizational Affiliation: