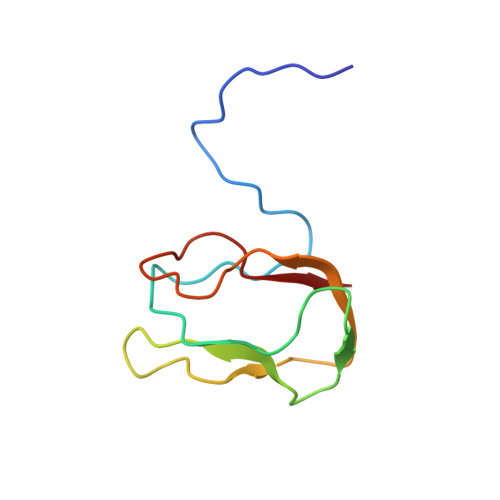
NMR Studies on the Solution Structure of a Deletion Mutant of the Transcarboxylase Biotin Carrier Subunit
Jank, M.M., Sadowsky, J.D., Peikert, C., Berger, S.(2002) Int J Biol Macromol 30: 233
- PubMed: 12297230 Search on PubMed
- DOI: https://doi.org/10.1016/s0141-8130(02)00033-8
- Primary Citation Related Structures:
1O78 - PubMed Abstract:
A deletion mutant of the transcarboxylase biotin carrier protein was completely labeled with 13C and 15N. A multitude of 2D and 3D NMR spectra were recorded and assigned. An NMR solution structure was derived from the data and compared in detail with the recently published structure of the wild-type. It is shown that deletion of 30% of the amino acids does not alter the structure of the rigid protein core.
- Institut für Analytische Chemie, Universität Leipzig, Linnestrasse 3, D-04103, Leipzig, Germany.
Organizational Affiliation: