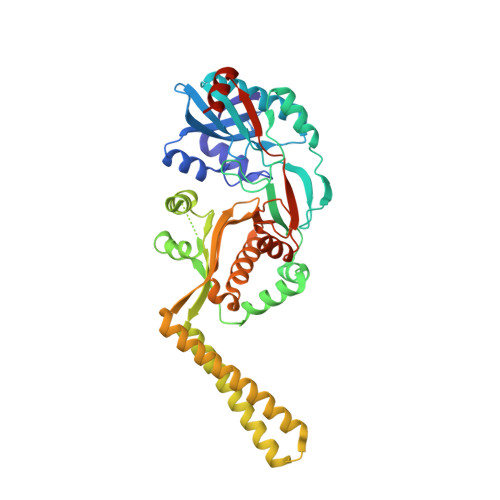
X-ray crystal structure of Staphylococcus aureus FemA.
Benson, T.E., Prince, D.B., Mutchler, V.T., Curry, K.A., Ho, A.M., Sarver, R.W., Hagadorn, J.C., Choi, G.H., Garlick, R.L.(2002) Structure 10: 1107-1115
- PubMed: 12176388 Search on PubMed
- DOI: https://doi.org/10.1016/s0969-2126(02)00807-9
- Primary Citation Related Structures:
1LRZ - PubMed Abstract:
The latter stages of peptidoglycan biosynthesis in Staphylococci involve the synthesis of a pentaglycine bridge on the epsilon amino group of the pentapeptide lysine side chain. Genetic and biochemical evidence suggest that sequential addition of these glycines is catalyzed by three homologous enzymes, FemX (FmhB), FemA, and FemB. The first protein structure from this family, Staphylococcus aureus FemA, has been solved at 2.1 A resolution by X-ray crystallography. The FemA structure reveals a unique organization of several known protein folds involved in peptide and tRNA binding. The surface of the protein also reveals an L-shaped channel suitable for a peptidoglycan substrate. Analysis of the structural features of this enzyme provides clues to the mechanism of action of S. aureus FemA.
- Structural, Analytical, and Medicinal Chemistry, Pharmacia Corporation, Kalamazoo, MI 49007, USA. timothy.e.benson@pharmacia.com
Organizational Affiliation: