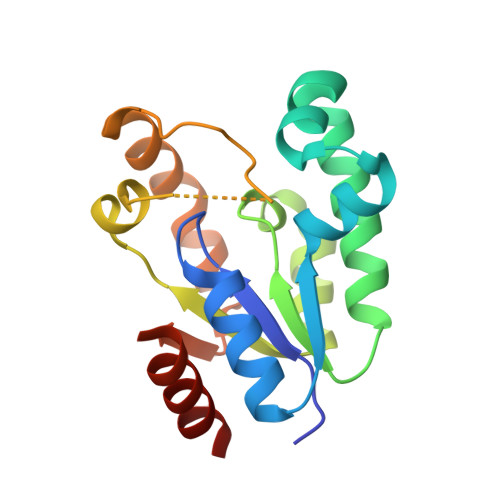
Crystal structure of the Escherichia coli shikimate kinase I (AroK) that confers sensitivity to mecillinam.
Romanowski, M.J., Burley, S.K.(2002) Proteins 47: 558-562
- PubMed: 12001235 Search on PubMed
- DOI: https://doi.org/10.1002/prot.10099
- Primary Citation Related Structures:
1KAG - Laboratories of Molecular Biophysics, The Rockefeller University, New York, New York.
Organizational Affiliation: