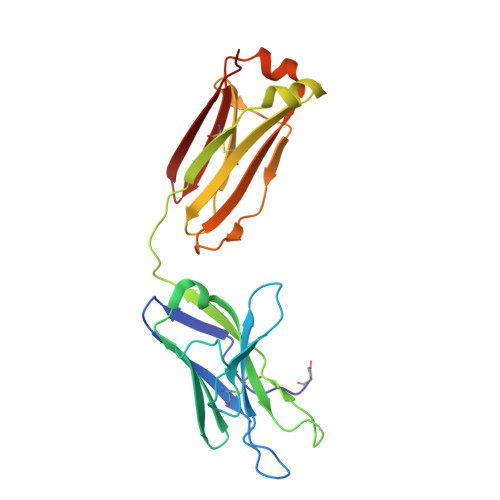
Three-dimensional structure of an immunoglobulin light-chain dimer with amyloidogenic properties.
Bourne, P.C., Ramsland, P.A., Shan, L., Fan, Z.C., DeWitt, C.R., Shultz, B.B., Terzyan, S.S., Moomaw, C.R., Slaughter, C.A., Guddat, L.W., Edmundson, A.B.(2002) Acta Crystallogr D Biol Crystallogr 58: 815-823
- PubMed: 11976493 Search on PubMed
- DOI: https://doi.org/10.1107/s0907444902004183
- Primary Citation Related Structures:
1JVK - PubMed Abstract:
The X-ray structure of an immunoglobulin light-chain dimer isolated from the urine as a "Bence-Jones protein" from a patient with multiple myeloma and amyloidosis (Sea) was determined at 1.94 A resolution and refined to R and R(free) factors of 0.22 and 0.25, respectively. This "amyloidogenic" protein crystallized in the orthorhombic P2(1)2(1)2(1) space group with unit-cell parameters a = 48.28, b = 83.32, c = 112.59 A as determined at 100 K. In the vital organs (heart and kidneys), the equivalent of the urinary protein produced fibrillar amyloid deposits which were fatal to the patient. Compared with the amyloidogenic Mcg light-chain dimer, the Sea protein was highly soluble in aqueous solutions and only crystallized at concentrations approaching 100 mg ml(-1). Both the Sea and Mcg proteins packed into crystals in highly ordered arrangements typical of strongly diffracting crystals of immunoglobulin fragments. Overall similarities and significant differences in the three-dimensional structures and crystalline properties are discussed for the Sea and Mcg Bence-Jones proteins, which together provide a generalized model of abnormalities present in lambda chains, facilitating a better understanding of amyloidosis of light-chain origin (AL).
- OMRF, 825 N E 13th Street, Oklahoma City, OK 73104, USA.
Organizational Affiliation: