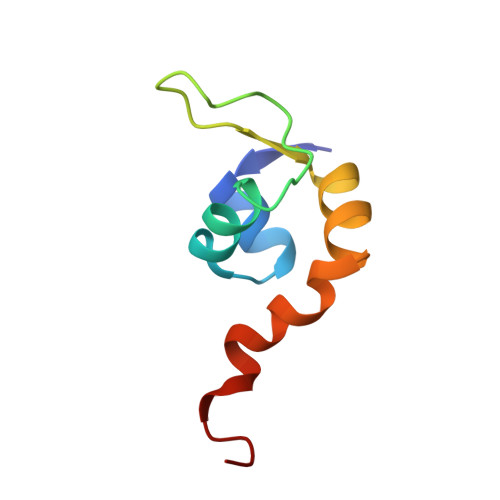
Insights into specific DNA recognition during the assembly of a viral genome packaging machine.
de Beer, T., Fang, J., Ortega, M., Yang, Q., Maes, L., Duffy, C., Berton, N., Sippy, J., Overduin, M., Feiss, M., Catalano, C.E.(2002) Mol Cell 9: 981-991
- PubMed: 12049735 Search on PubMed
- DOI: https://doi.org/10.1016/s1097-2765(02)00537-3
- Primary Citation Related Structures:
1J9I - PubMed Abstract:
Terminase enzymes mediate genome "packaging" during the reproduction of DNA viruses. In lambda, the gpNu1 subunit guides site-specific assembly of terminase onto DNA. The structure of the dimeric DNA binding domain of gpNu1 was solved using nuclear magnetic resonance spectroscopy. Its fold contains a unique winged helix-turn-helix (wHTH) motif within a novel scaffold. Surprisingly, a predicted P loop ATP binding motif is in fact the wing of the DNA binding motif. Structural and genetic analysis has identified determinants of DNA recognition specificity within the wHTH motif and the DNA recognition sequence. The structure reveals an unexpected DNA binding mode and provides a mechanistic basis for the concerted action of gpNu1 and Escherichia coli integration host factor during assembly of the packaging machinery.
- Department of Pharmacology, University of Colorado Health Sciences Center, Denver 80262, USA. tonny.debeer@astrazeneca.com
Organizational Affiliation: