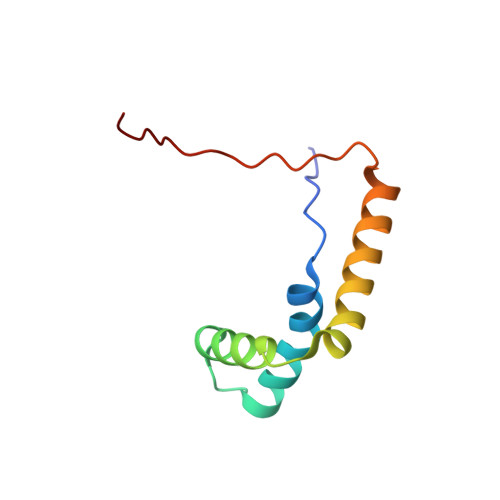
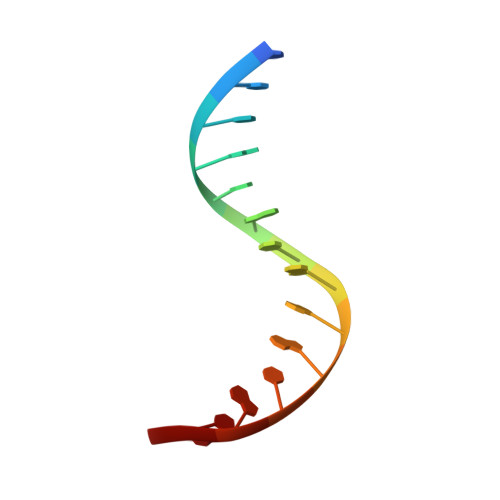
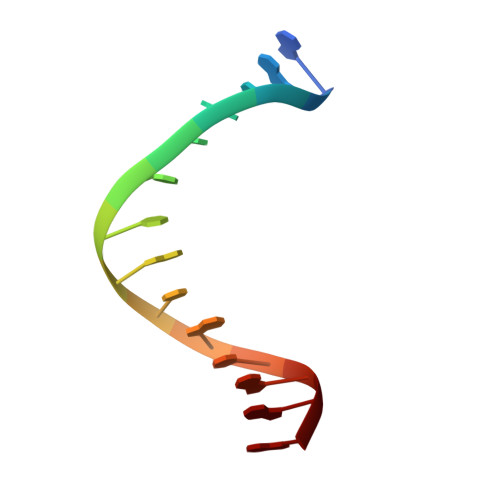
Structural basis for SRY-dependent 46-X,Y sex reversal: modulation of DNA bending by a naturally occurring point mutation.
Murphy, E.C., Zhurkin, V.B., Louis, J.M., Cornilescu, G., Clore, G.M.(2001) J Mol Biology 312: 481-499
- PubMed: 11563911 Search on PubMed
- DOI: https://doi.org/10.1006/jmbi.2001.4977
- Primary Citation Related Structures:
1J46, 1J47 - PubMed Abstract:
The HMG-box domain of the human male sex-determining factor SRY, hSRY(HMG) (comprising residues 57-140 of the full-length sequence), binds DNA sequence-specifically in the minor groove, resulting in substantial DNA bending. The majority of point mutations resulting in 46X,Y sex reversal are located within this domain. One clinical de novo mutation, M64I in the full-length hSRY sequence, which corresponds to M9I in the present hSRY(HMG) construct, acts principally by reducing the extent of DNA bending. To elucidate the structural consequences of the M9I mutation, we have solved the 3D solution structures of wild-type and M9I hSRY(HMG) complexed to a DNA 14mer by NMR, including the use of residual dipolar couplings to derive long-range orientational information. We show that the average bend angle (derived from an ensemble of 400 simulated annealing structures for each complex) is reduced by approximately 13 degrees from 54(+/-2) degrees in the wild-type complex to 41(+/-2) degrees in the M9I complex. The difference in DNA bending can be localized directly to changes in roll and tilt angles in the ApA base-pair step involved in interactions with residue 9 and partial intercalation of Ile13. The larger bend angle in the wild-type complex arises as a direct consequence of steric repulsion of the sugar of the second adenine by the bulky S(delta) atom of Met9, whose position is fixed by a hydrogen bond with the guanidino group of Arg17. In the M9I mutant, this hydrogen bond can no longer occur, and the less bulky C(gamma)m methyl group of Ile9 braces the sugar moieties of the two adenine residues, thereby decreasing the roll and tilt angles at the ApA step by approximately 8 degrees and approximately 5 degrees, respectively, and resulting in an overall difference in bend angle of approximately 13 degrees between the two complexes. To our knowledge, this is one of the first examples where the effects of a clinical mutation involving a protein-DNA complex have been visualized at the atomic level.
- Laboratory of Chemical Physics, Building 5, National Institute of Diabetes and Digestive and Kidney Diseases, National Institutes of Health, Bethesda, MD 20892-0510, USA.
Organizational Affiliation: