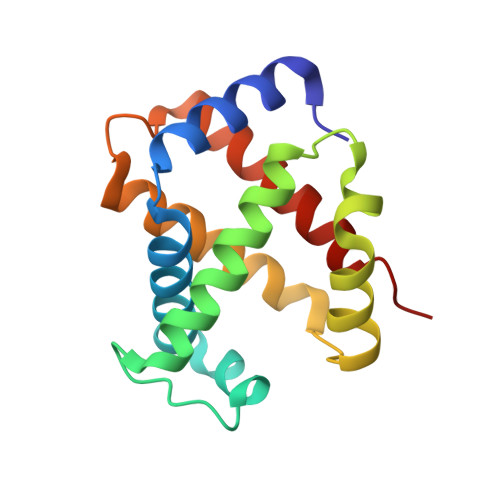
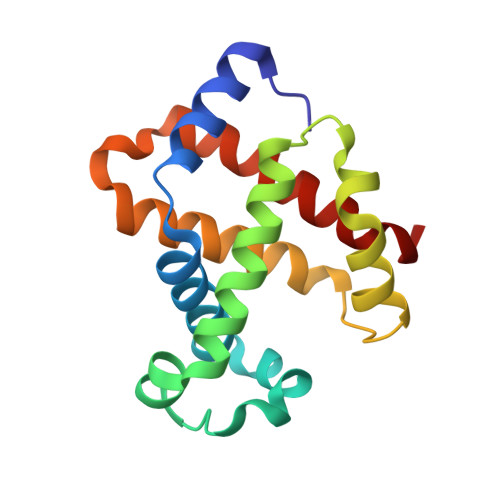
Crystal Structure of Horse Carbonmonoxyhemoglobin-Bezafibrate Complex at 1.55A Resolution. A Novel Allosteric Binding Site in R-State Hemoglobin
Shibayama, N., Miura, S., Tame, J.R.H., Yonetani, T., Park, S.-Y.(2002) J Biological Chem 277: 38791-38796
- PubMed: 12122004 Search on PubMed
- DOI: https://doi.org/10.1074/jbc.M205461200
- Primary Citation Related Structures:
1IWH - PubMed Abstract:
Bezafibrate, an antilipidemic drug, is known as a potent allosteric effector of hemoglobin. The previously proposed mechanism for the allosteric potency of this drug was that it stabilizes and constrains the T-state of hemoglobin by specifically binding to the large central cavity of the T-state. Here we report a new allosteric binding site of fully liganded R-state hemoglobin for this drug. The high resolution crystal structure of horse carbonmonoxyhemoglobin in complex with bezafibrate reveals that the bezafibrate molecule lies near the surface of the E-helix of each alpha subunit and the complex maintains the quaternary structure of the R-state. Binding is caused by the close fit of bezafibrate into the binding pocket, which is composed of some hydrophobic residues and the heme edge, suggesting the importance of hydrophobic interactions. Upon binding of bezafibrate, the distance between Fe and the N epsilon(2) of distal His E7(alpha 58) is shortened by 0.22 A in the alpha subunit, whereas no significant structural changes are transmitted to the beta subunit. Oxygen equilibrium studies of R-state-locked hemoglobin with bezafibrate in a wet porous sol-gel indicate that bezafibrate selectively lowers the oxygen affinity of one type of subunit within the R-state, consistent with the structural data. These results disclose a new allosteric mechanism of bezafibrate and offer the first demonstration of how the allosteric effector interacts with R-state hemoglobin.
- Department of Physiology, Division of Biophysics, Jichi Medical School, Yakushiji 3311-1, Minamikawachi, Kawachi, Tochigi 329-0498, Japan. shibayam@jichi.ac.jp
Organizational Affiliation: