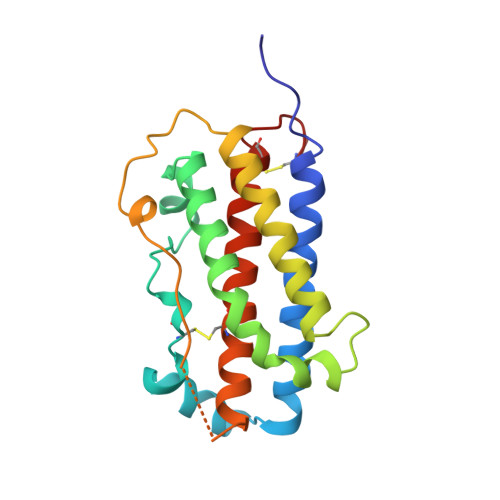
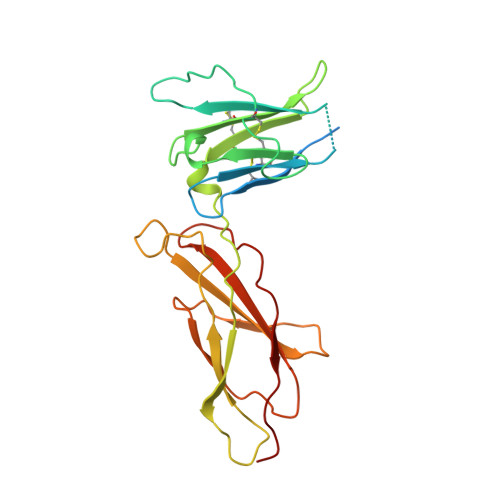
Crystal structure of an antagonist mutant of human growth hormone, G120R, in complex with its receptor at 2.9 A resolution.
Sundstrom, M., Lundqvist, T., Rodin, J., Giebel, L.B., Milligan, D., Norstedt, G.(1996) J Biological Chem 271: 32197-32203
- PubMed: 8943276 Search on PubMed
- DOI: https://doi.org/10.1074/jbc.271.50.32197
- Primary Citation Related Structures:
1HWG, 1HWH - PubMed Abstract:
Human growth hormone binds two receptor molecules and thereby induces signal transduction through receptor dimerization. At high concentrations, growth hormone acts as an antagonist because of a large difference in affinities at the respective binding sites. This antagonist action can be enhanced further by reducing binding in the low affinity binding site. A growth hormone antagonist mutant Gly-120 --> Arg, has been crystallized with its receptor as a 1:1 complex and the crystal structure determined at 2.9 A resolution. The 1:1 complex is remarkably similar to the native growth hormone-receptor 1:2 complex. A comparison between the two structures reveals only minimal differences in the conformations of the hormone or its receptor in the two complexes, including the angle between the two immunoglobulin-like domains of the receptor. Further, two symmetry-related 1:1 complexes in the crystal form a 2:2 complex with a large solvent inaccessible area between two receptor molecules. In addition, we present here a native human growth hormone-human growth hormone-binding protein 1:2 complex structure at 2.5 A resolution. One important difference between our structure and the previously published crystal structure at 2.8 A is revealed. Trp-104 in the receptor, a key residue in the hormone-receptor interaction, has an altered conformation in the low affinity site enabling a favorable hydrogen bond to be formed with Asp-116 of the hormone.
- Department of Structural Biochemistry, Pharmacia and Upjohn, Preclinical Research and Development, S-112 87 Stockholm, Sweden. michael.sundstrom@sto.pharmacia.se
Organizational Affiliation: