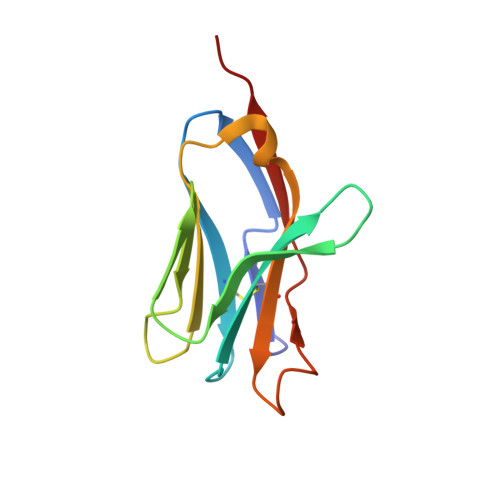
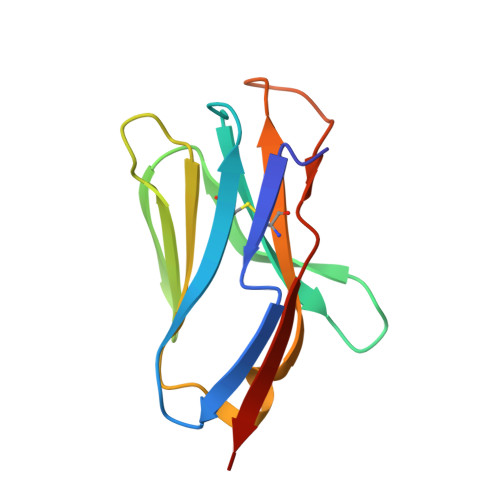
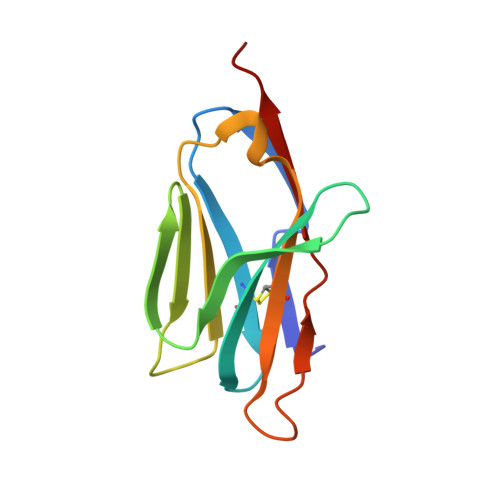
Crystal Structure of a T Cell Receptor Valpha11 (Av11S5) Domain: New Canonical Forms for the First and Second Complementarity Determining Regions
Machius, M., Cianga, P., Deisenhofer, J., Ward, E.S.(2001) J Mol Biology 310: 689
- PubMed: 11453680
- DOI: https://doi.org/10.1006/jmbi.2001.4794
- Primary Citation of Related Structures:
1H5B - PubMed Abstract:
We describe the X-ray crystallographic structure of a murine T cell receptor (TCR) Valpha domain ("Valpha85.33"; AV11S5-AJ17) to 1.85 A resolution. The Valpha85.33 domain is derived from a TCR that recognizes a type II collagen peptide associated with the murine major histocompatibility complex (MHC) class II molecule, I-A(q). Valpha85.33 packs as a Valpha-Valpha homodimer with a highly symmetric monomer-monomer interface. The first and second complementarity determining regions (CDR1 and CDR2) of this Valpha are shorter than the CDRs corresponding to the majority of other Valpha gene families, and three-dimensional structures of CDRs of these lengths have not been described previously. The CDR1 and CDR2 therefore represent new canonical forms that could serve as templates for AV11 family members. CDR3 of the Valpha85.33 domain is highly flexible and this is consistent with plasticity of this region of the TCR. The fourth hypervariable loop (HV4alpha) of AV11 and AV10 family members is one residue longer than that of other HV4alpha regions and shows a high degree of flexibility. The increase in length results in a distinct disposition of the conserved residue Lys68, which has been shown in other studies to play a role in antigen recognition. The X-ray structure of Valpha85.33 extends the database of canonical forms for CDR1 and CDR2, and has implications for antigen recognition by TCRs that contain related Valpha domains.
- Howard Hughes Medical Institute and Department of Biochemistry, University of Texas Southwestern Medical Center, 5323 Harry Hines Blvd., Dallas, TX 75390-9050, USA.
Organizational Affiliation: