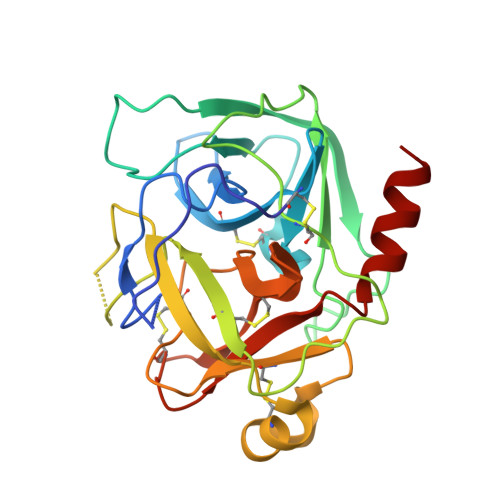
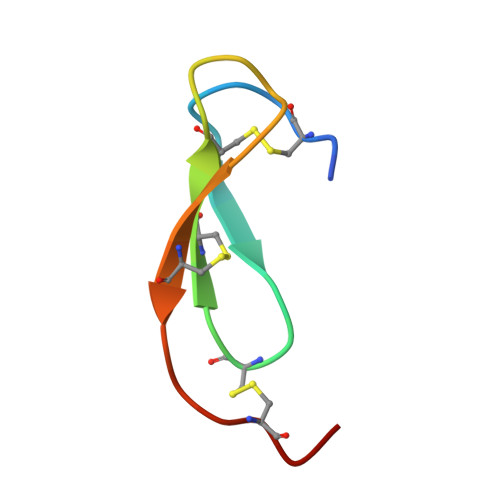
Complexation of Two Proteic Insect Inhibitors to the Active Site of Chymotrypsin Suggests Decoupled Roles for Binding and Selectivity
Roussel, A., Mathieu, M., Dobbs, A., Luu, B., Cambillau, C., Kellenberger, C.(2001) J Biological Chem 276: 38893
- PubMed: 11495915 Search on PubMed
- DOI: https://doi.org/10.1074/jbc.M105707200
- Primary Citation Related Structures:
1GL0, 1GL1 - PubMed Abstract:
The crystal structures of two homologous inhibitors (PMP-C and PMP-D2v) from the insect Locusta migratoria have been determined in complex with bovine alpha-chymotrypsin at 2.1- and 3.0-A resolution, respectively. PMP-C is a potent bovine alpha-chymotrypsin inhibitor whereas native PMP-D2 is a weak inhibitor of bovine trypsin. One unique mutation at the P1 position converts PMP-D2 into a potent bovine alpha-chymotrypsin inhibitor. The two peptides have a similar overall conformation, which consists of a triple-stranded antiparallel beta-sheet connected by three disulfide bridges, thus defining a novel family of serine protease inhibitors. They have in common the protease interaction site, which is composed of the classical protease binding loop (position P5 to P'4, corresponding to residues 26-34) and of an internal segment (residues 15-18), held together by two disulfide bridges. Structural divergences between the two inhibitors result in an additional interaction site between PMP-D2v (position P10 to P6, residues 21-25) and the residues 172-175 of alpha-chymotrypsin. This unusual interaction may be responsible for species selectivity. A careful comparison of data on bound and free inhibitors (from this study and previous NMR studies, respectively) suggests that complexation to the protease stabilizes the flexible binding loop (from P5 to P'4).
- Architecture et Fonction de Macromolecules Biologiques, UMR-6098, CNRS et Universités d'Aix-Marseille I et II, 31 chemin Joseph Aiguier, 13402 Marseille Cedex 20, France. roussel@afmb.cnrs-mrs.fr
Organizational Affiliation: