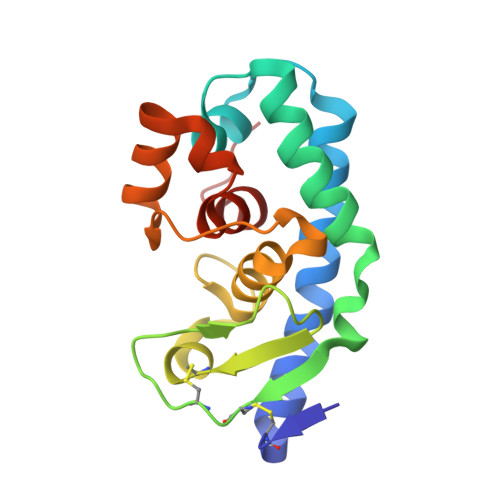
Structure of a new 'aspzincin' metalloendopeptidase from Grifola frondosa: implications for the catalytic mechanism and substrate specificity based on several different crystal forms.
Hori, T., Kumasaka, T., Yamamoto, M., Nonaka, N., Tanaka, N., Hashimoto, Y., Ueki, U., Takio, K.(2001) Acta Crystallogr D Biol Crystallogr 57: 361-368
- PubMed: 11223512 Search on PubMed
- DOI: https://doi.org/10.1107/s0907444900019740
- Primary Citation Related Structures:
1G12, 1GE5, 1GE6, 1GE7 - PubMed Abstract:
Crystal structures of a peptidyl-Lys metalloendopeptidase (MEP) from the edible mushroom Grifola frondosa (GfMEP) were solved in four crystal forms. This represents the first structure of the new family 'aspzincins' with a novel active-site architecture. The active site is composed of two helices and a loop region and includes the HExxH and GTxDxxYG motifs conserved among aspzincins. His117, His121 and Asp130 coordinate to the catalytic zinc ligands. An electrostatically negative region composed of Asp154 and Glu157 attracts a positively charged Lys side chain of a substrate in a specific manner. A Tyr133 side chain located on the S1' pocket had different configurations in two crystal forms and was not observed in the other crystal forms. The flexible Tyr133 plays two roles in the enzymatic function of GfMEP. The first is to provide a hydrophobic environment with Phe83 in order to accommodate the alkyl part of the Lys side chain of a substrate and the second is as a 'proton donor' to the oxyanion of the tetrahedral transition state to stabilize the reaction transition state.
- RIKEN Harima Institute, 1-1-1 Kouto, Mikazuki, Sayo-gun 679-5148, Japan.
Organizational Affiliation: