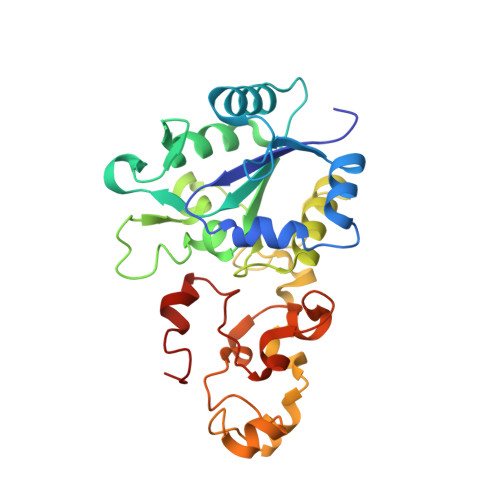
3Fe-4S] to [4Fe-4S] cluster conversion in Desulfovibrio fructosovorans [NiFe] hydrogenase by site-directed mutagenesis
Rousset, M., Montet, Y., Guigliarelli, B., Forget, N., Asso, M., Bertrand, P., Fontecilla-Camps, J.C., Hatchikian, E.C.(1998) Proc Natl Acad Sci U S A 95: 11625-11630
- PubMed: 9751716
- DOI: https://doi.org/10.1073/pnas.95.20.11625
- Primary Citation of Related Structures:
1FRF - PubMed Abstract:
The role of the high potential [3Fe-4S]1+,0 cluster of [NiFe] hydrogenase from Desulfovibrio species located halfway between the proximal and distal low potential [4Fe-4S]2+,1+ clusters has been investigated by using site-directed mutagenesis. Proline 238 of Desulfovibrio fructosovorans [NiFe] hydrogenase, which occupies the position of a potential ligand of the lacking fourth Fe-site of the [3Fe-4S] cluster, was replaced by a cysteine residue. The properties of the mutant enzyme were investigated in terms of enzymatic activity, EPR, and redox properties of the iron-sulfur centers and crystallographic structure. We have shown on the basis of both spectroscopic and x-ray crystallographic studies that the [3Fe-4S] cluster of D. fructosovorans hydrogenase was converted into a [4Fe-4S] center in the P238 mutant. The [3Fe-4S] to [4Fe-4S] cluster conversion resulted in a lowering of approximately 300 mV of the midpoint potential of the modified cluster, whereas no significant alteration of the spectroscopic and redox properties of the two native [4Fe-4S] clusters and the NiFe center occurred. The significant decrease of the midpoint potential of the intermediate Fe-S cluster had only a slight effect on the catalytic activity of the P238C mutant as compared with the wild-type enzyme. The implications of the results for the role of the high-potential [3Fe-4S] cluster in the intramolecular electron transfer pathway are discussed.
- Unité de Bioénergétique et Ingéniérie des Protéines, Institut de Biologie Structurale et Microbiologie, Centre National de la Recherche Scientifique, 31, Chemin Joseph Aiguier, 13402 Marseille CDX 20, France.
Organizational Affiliation: