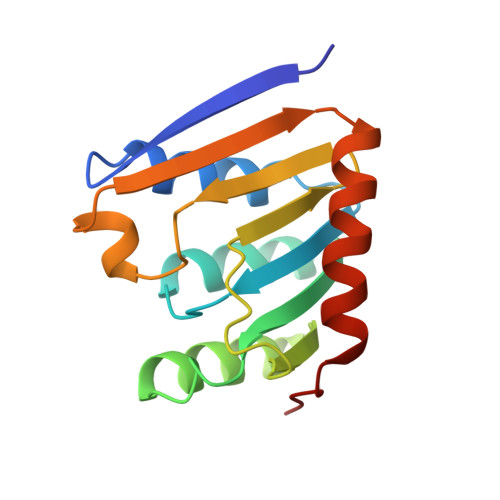
Crystal structure of a phospholipase D family member.
Stuckey, J.A., Dixon, J.E.(1999) Nat Struct Biol 6: 278-284
- PubMed: 10074947
- DOI: https://doi.org/10.1038/6716
- Primary Citation Related Structures:
1BYR, 1BYS - PubMed Abstract:
The first crystal structure of a phospholipase D (PLD) family member has been determined at 2.0 A resolution. The PLD superfamily is defined by a common sequence motif, HxK(x)4D(x)6GSxN, and includes enzymes involved in signal transduction, lipid biosynthesis, endonucleases and open reading frames in pathogenic viruses and bacteria. The crystal structure suggests that residues from two sequence motifs form a single active site. A histidine residue from one motif acts as a nucleophile in the catalytic mechanism, forming a phosphoenzyme intermediate, whereas a histidine residue from the other motif appears to function as a general acid in the cleavage of the phosphodiester bond. The structure suggests that the conserved lysine residues are involved in phosphate binding. Large-scale genomic sequencing revealed that there are many PLD family members. Our results suggest that all of these proteins may possess a common structure and catalytic mechanism.
- Department of Biological Chemistry, The University of Michigan, Ann Arbor 48109-0606, USA.
Organizational Affiliation: