Crystal structure of Thermoactinomyces vulgaris R-47 alpha-amylase II (TVAII) hydrolyzing cyclodextrins and pullulan at 2.6 A resolution.
Kamitori, S., Kondo, S., Okuyama, K., Yokota, T., Shimura, Y., Tonozuka, T., Sakano, Y.(1999) J Mol Biology 287: 907-921
- PubMed: 10222200
- DOI: https://doi.org/10.1006/jmbi.1999.2647
- Primary Citation Related Structures:
1BVZ - PubMed Abstract:
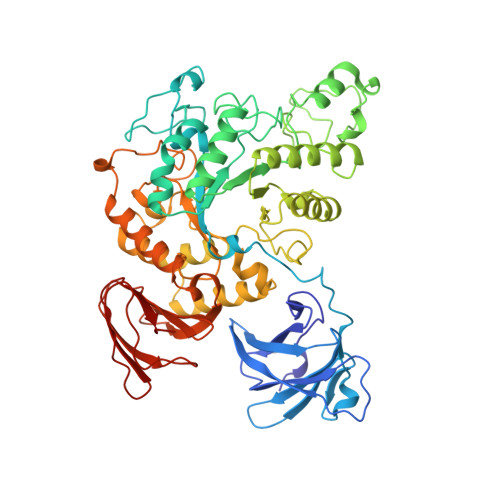
The crystal structure of Thermoactinomyces vulgaris R-47 alpha-Amylase II (TVAII) has been determined by multiple isomorphous replacement at 2.6 A resolution. TVAII was crystallized in an orthorhombic system with the space group P212121 and the cell dimensions a=118.5 A, b=119.5 A, c=114.5 A. There are two molecules in an asymmetric unit, related by the non-crystallographic 2-fold symmetry. Diffraction data were collected at 113 K and the cell dimensions reduced to a=114.6 A, b=117.9 A, c=114.2 A, and the model was refined against 7.0-2.6 A resolution data giving an R-factor of 0.204 (Rfree=0.272). The final model consists of 1170 amino acid residues (two molecules) and 478 water molecules with good chemical geometry. TVAII has three domains, A, B, and C, like other alpha-amylases. Domain A with a (beta/alpha)8 barrel structure and domain C with a beta-sandwich structure are very similar to those found in other alpha-amylases. Additionally, TVAII has an extra domain N composed of 121 amino acid residues at the N-terminal site, which has a beta-barrel-like structure consisting of seven antiparallel beta-strands. Domain N is one of the driving forces in the formation of the dimer structure of TVAII, but its role in the enzyme activity is still not clear. TVAII does not have the Ca2+ binding site that connects domains A and B in other alpha-amylases, rather the NZ atom of Lys299 of TVAII serves as the connector between these domains. TVAII can hydrolyze cyclodextrins and pullulan as well as starch. Based on a structural comparison with the complex between a mutant cyclodextrin glucanotransferase and a beta-cyclodextrin derivative, Phe286 located at domain B is considered the residue most likely to recognize the hydrophobic cavity of cyclodextrins. The active-site cleft of TVAII is wider and shallower than that of other alpha-amylases, and seems to be suitable for the binding of pullulan which is expected not to adopt the helical structure of amylose.
- Department of Biotechnology and Life Science, Faculty of Technology, Tokyo University of Agriculture and Technology, Koganei, Tokyo, 184-8588, Japan.kamitori@cc.tuat.ac.jp
Organizational Affiliation: