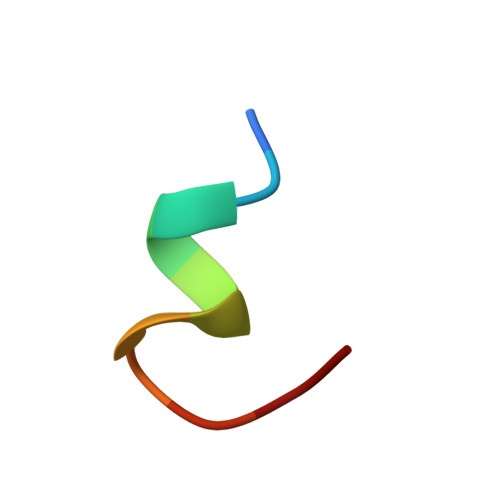Light-activated rhodopsin induces structural binding motif in G protein alpha subunit.
Kisselev, O.G., Kao, J., Ponder, J.W., Fann, Y.C., Gautam, N., Marshall, G.R.(1998) Proc Natl Acad Sci U S A 95: 4270-4275
- PubMed: 9539726
- DOI: https://doi.org/10.1073/pnas.95.8.4270
- Primary Citation Related Structures:
1AQG - PubMed Abstract:
A large superfamily of transmembrane receptors control cellular responses to diverse extracellular signals by catalyzing activation of specific types of heterotrimeric GTP-binding proteins. How these receptors recognize and promote nucleotide exchange on G protein alpha subunits to initiate signal amplification is unknown. The three-dimensional structure of the transducin (Gt) alpha subunit C-terminal undecapeptide Gtalpha(340-350) IKENLKDCGLF was determined by transferred nuclear Overhauser effect spectroscopy while it was bound to photoexcited rhodopsin. Light activation of rhodopsin causes a dramatic shift from a disordered conformation of Gtalpha(340-350) to a binding motif with a helical turn followed by an open reverse turn centered at Gly-348, a helix-terminating C capping motif of an alphaL type. Docking of the NMR structure to the GDP-bound x-ray structure of Gt reveals that photoexcited rhodopsin promotes the formation of a continuous helix over residues 325-346 terminated by the C-terminal helical cap with a unique cluster of crucial hydrophobic side chains. A molecular mechanism by which activated receptors can control G proteins through reversible conformational changes at the receptor-G protein interface is demonstrated.
- Institute for Biomedical Computing, Washington University Medical School, St. Louis, MO 63110, USA.
Organizational Affiliation: