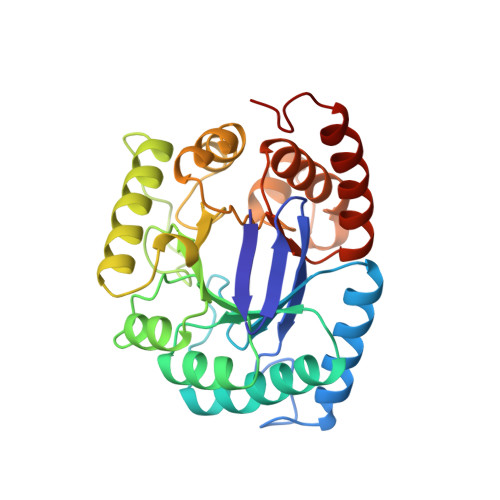
Crystal structure of the anti-bacterial sulfonamide drug target dihydropteroate synthase.
Achari, A., Somers, D.O., Champness, J.N., Bryant, P.K., Rosemond, J., Stammers, D.K.(1997) Nat Struct Biol 4: 490-497
- PubMed: 9187658 Search on PubMed
- DOI: https://doi.org/10.1038/nsb0697-490
- Primary Citation Related Structures:
1AJ0, 1AJ2, 1AJZ - PubMed Abstract:
Sulfonamides were amongst the first clinically useful antibacterial agents to be discovered. The identification of sulfanilamide as the active component of the dye Prontosil rubrum led to the synthesis of clinically useful analogues. Today sulfamethoxazole (in combination with trimethoprim), is used to treat urinary tract infections caused by bacteria such as Escherichia coli and is also a first-line treatment for pneumonia caused by the fungus Pneumocystis carinii, a common condition in AIDS patients. The site of action is the de novo folate biosynthesis enzyme dihydropteroate synthase (DHPS) where sulfonamides act as analogues of one of the substrates, para-aminobenzoic acid (pABA). We report here the crystal structure of E.coli DHPS at 2.0 A resolution refined to an R-factor of 0.185. The single domain of 282 residues forms an eight-stranded alpha/beta-barrel. The 7,8-dihydropterin pyrophosphate (DHPPP) substrate binds in a deep cleft in the barrel, whilst sulfanilamide binds closer to the surface. The DHPPP ligand site is highly conserved amongst prokaryotic and eukaryotic DHPSs.
- Glaxo Wellcome Research & Development, Beckenham, Kent, UK. aa37795@ggr.co.uk
Organizational Affiliation: