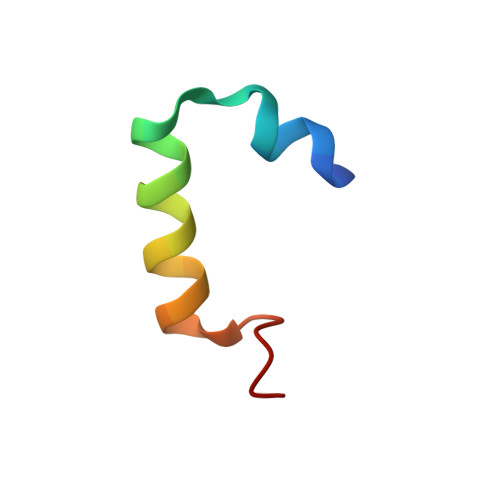
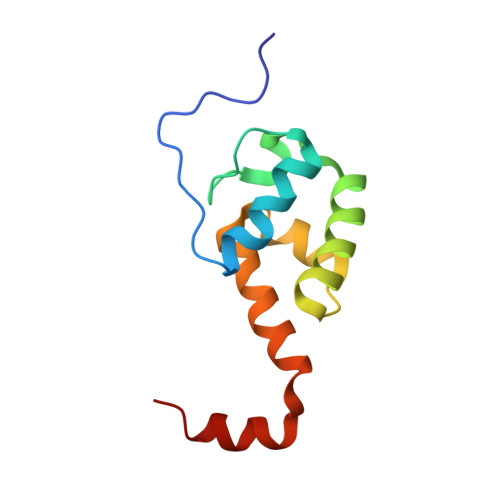
Cooperation between a T Domain and a Minimal C-Terminal Docking Domain to Enable Specific Assembly in a Multiprotein NRPS.
Watzel, J., Duchardt-Ferner, E., Sarawi, S., Bode, H.B., Wohnert, J.(2021) Angew Chem Int Ed Engl 60: 14171-14178
- PubMed: 33876501
- DOI: https://doi.org/10.1002/anie.202103498
- Primary Citation of Related Structures:
7B2B, 7B2F - PubMed Abstract:
Non-ribosomal peptide synthetases (NRPS) produce natural products from amino acid building blocks. They often consist of multiple polypeptide chains which assemble in a specific linear order via specialized N- and C-terminal docking domains ( N/C DDs). Typically, docking domains function independently from other domains in NRPS assembly. Thus, docking domain replacements enable the assembly of "designer" NRPS from proteins that normally do not interact. The multiprotein "peptide-antimicrobial-Xenorhabdus" (PAX) peptide-producing PaxS NRPS is assembled from the three proteins PaxA, PaxB and PaxC. Herein, we show that the small C DD of PaxA cooperates with its preceding thiolation (T 1 ) domain to bind the N DD of PaxB with very high affinity, establishing a structural and thermodynamical basis for this unprecedented docking interaction, and we test its functional importance in vivo in a truncated PaxS assembly line. Similar docking interactions are apparently present in other NRPS systems.
- Molecular Biotechnology, Institute of Molecular Biosciences, Goethe University Frankfurt, 60438, Frankfurt am Main, Germany.
Organizational Affiliation: