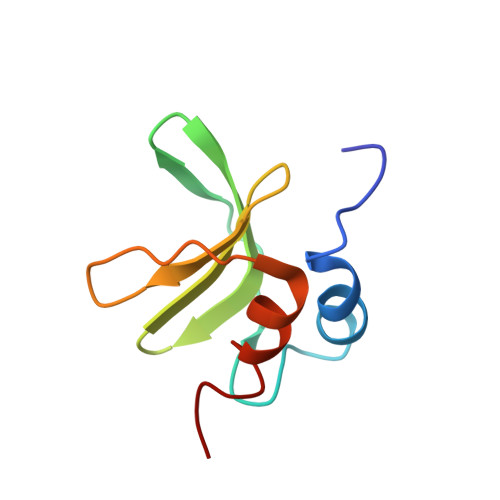
Structure of the SARS-Unique Domain C From the Bat Coronavirus HKU4.
Staup, A.J., De Silva, I.U., Catt, J.T., Tan, X., Hammond, R.G., Johnson, M.A.(2019) Nat Prod Commun 14: 1934578X19849202-1934578X19849202
- PubMed: 32395093
- DOI: https://doi.org/10.1177/1934578X19849202
- Primary Citation of Related Structures:
6MWM - PubMed Abstract:
Coronaviruses (CoVs) that cause infections such as severe acute respiratory syndrome (SARS) and Middle East respiratory syndrome phylogenetically originate from bat CoVs. The coronaviral nonstructural protein 3 (nsp3) has been implicated in viral replication, polyprotein cleavage, and host immune interference. We report the structure of the C domain from the SARS-Unique Domain of bat CoV HKU4. The protein has a frataxin fold, consisting of 5 antiparallel β strands packed against 2 α helices. Bioinformatics analyses and nuclear magnetic resonance experiments were conducted to investigate the function of HKU4 C. The results showed that HKU4 C engages in protein-protein interactions with the nearby M domain of nsp3. The HKU4 C residues involved in protein-protein interactions are conserved in group 2c CoVs, indicating a conserved function.
- Department of Chemistry, 1720 2nd Avenue S. CHEM 201, University of Alabama at Birmingham, AL, USA.
Organizational Affiliation: