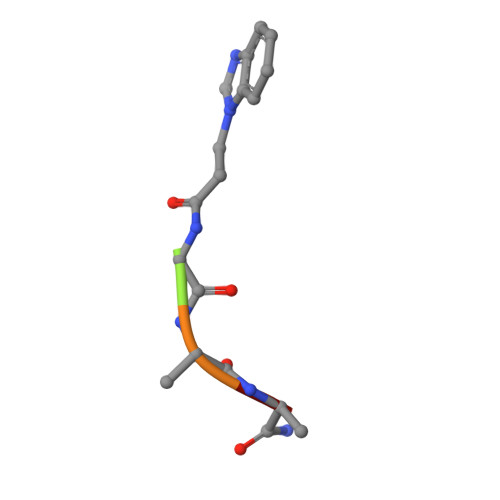
Replacement of an electron transfer pathway in cytochrome c peroxidase with a surrogate peptide
Hays Putnam, A.M., Lee, Y.T., Goodin, D.B.(2009) Biochemistry 48: 1-3
- PubMed: 19072042
- DOI: https://doi.org/10.1021/bi8020263
- Primary Citation of Related Structures:
3EXB - PubMed Abstract:
A proposed electron transfer pathway in cytochrome c peroxidase was previously excised from the structure by design. The engineered channel mutant was shown to bind peptide surrogates without restoration of cyt c oxidation. Here, we report the 1.6 A crystal structure of (N-benzimidazole-propionic acid)-Gly-Ala-Ala bound within the engineered channel. The peptide retains many features of the native electron transfer pathway: placement of benzimidazole at the position of the Trp-191 radical, hydrogen bonding to Asp235, and positioning of the C-terminus near the point where wild type CcP makes closest contact to cyt c. The inability of this surrogate pathway to restore function supports proposals that electron transfer requires the Trp-191 radical.
- Department of Molecular Biology, The Scripps Research Institute, 10550 North Torrey Pines Road, La Jolla, California 92037, USA.
Organizational Affiliation: