Fortuitous structure determination of 'as-isolated' Escherichia coli bacterioferritin in a novel crystal form.
van Eerde, A., Wolterink-van Loo, S., van der Oost, J., Dijkstra, B.W.(2006) Acta Crystallogr Sect F Struct Biol Cryst Commun 62: 1061-1066
- PubMed: 17077480
- DOI: https://doi.org/10.1107/S1744309106039583
- Primary Citation of Related Structures:
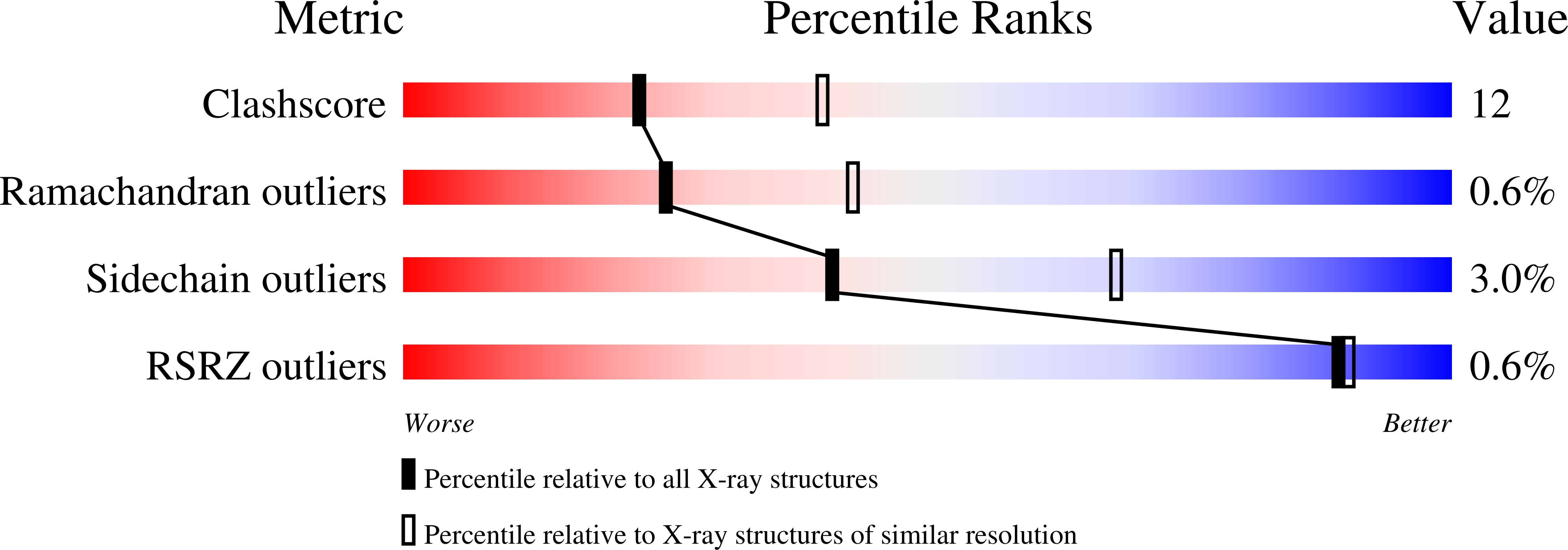
2HTN - PubMed Abstract:
Escherichia coli bacterioferritin was serendipitously crystallized in a novel cubic crystal form and its structure could be determined to 2.5 A resolution despite a high degree of merohedral twinning. This is the first report of crystallographic data on 'as-isolated' E. coli bacterioferritin. The ferroxidase active site contains positive difference density consistent with two metal ions that had co-purified with the protein. X-ray fluorescence studies suggest that the metal composition is different from that of previous structures and is a mix of zinc and native iron ions. The ferroxidase-centre configuration displays a similar flexibility as previously noted for other bacterioferritins.
Organizational Affiliation:
Laboratory of Biophysical Chemistry, University of Groningen, Nijenborgh 4, 9747 AG Groningen, The Netherlands.