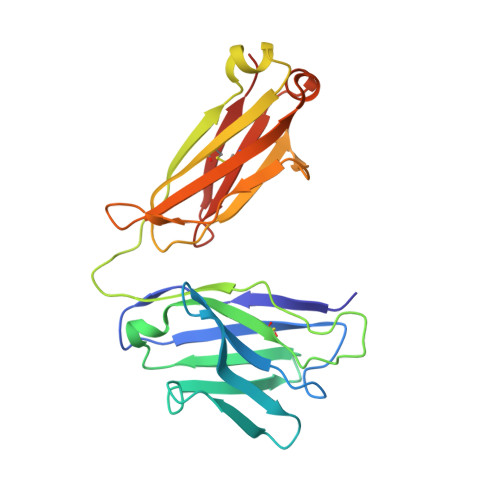
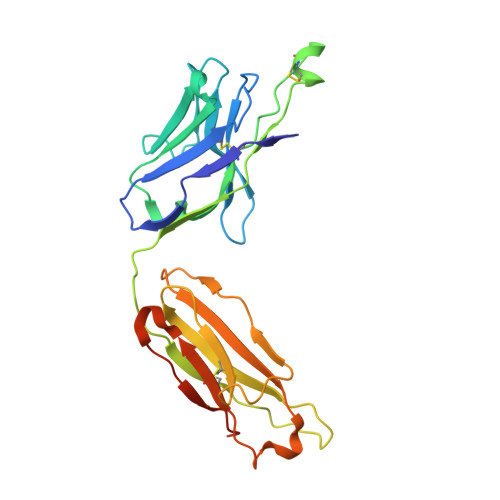
Bringing the Heavy Chain to Light: Creating a Symmetric, Bivalent IgG-Like Bispecific.
Ramasubramanian, A., Tennyson, R., Magnay, M., Kathuria, S., Travaline, T., Jain, A., Lord, D.M., Salemi, M., Sullivan, C., Magnay, T., Hu, J., Bric-Furlong, E., Rival, P., Zhou, Y., Hoffmann, D., Brondyk, W., Radosevic, K., Chowdhury, P.S.(2020) Antibodies (Basel) 9
- PubMed: 33172091
- DOI: https://doi.org/10.3390/antib9040062
- Primary Citation of Related Structures:
7JKB - PubMed Abstract:
Bispecific molecules are biologically significant, yet their complex structures pose important manufacturing and pharmacokinetic challenges. Nevertheless, owing to similarities with monoclonal antibodies (mAbs), IgG-like bispecifics conceptually align well with conventional expression and manufacturing platforms and often exhibit potentially favorable drug metabolism and pharmacokinetic (DMPK) properties. However, IgG-like bispecifics do not possess target bivalency and current designs often require tedious engineering and purification to ensure appropriate chain pairing. Here, we present a near-native IgG antibody format, the 2xVH, which can create bivalency for each target or epitope and requires no engineering for cognate chain pairing. In this modality, two different variable heavy (VH) domains with distinct binding specificities are grafted onto the first constant heavy (CH1) and constant light (CL) domains, conferring the molecule with dual specificity. To determine the versatility of this format, we characterized the expression, binding, and stability of several previously identified soluble human VH domains. By grafting these domains onto an IgG scaffold, we generated several prototype 2xVH IgG and Fab molecules that display similar properties to mAbs. These molecules avoided the post-expression purification necessary for engineered bispecifics while maintaining a capacity for simultaneous dual binding. Hence, the 2xVH format represents a bivalent, bispecific design that addresses limitations of manufacturing IgG-like bispecifics while promoting biologically-relevant dual target engagement.
Organizational Affiliation:
Biologics Research, Sanofi R&D, 49 New York Avenue, Framingham, MA 01701, USA.