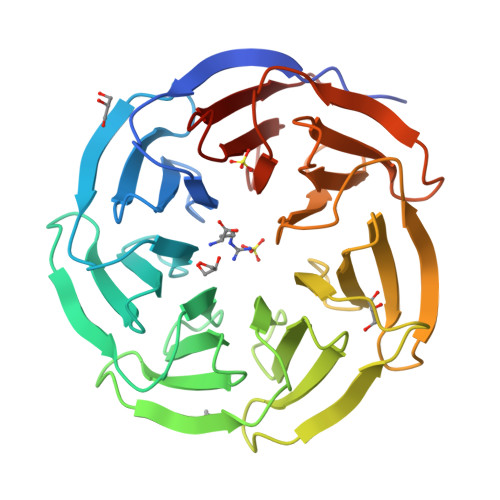
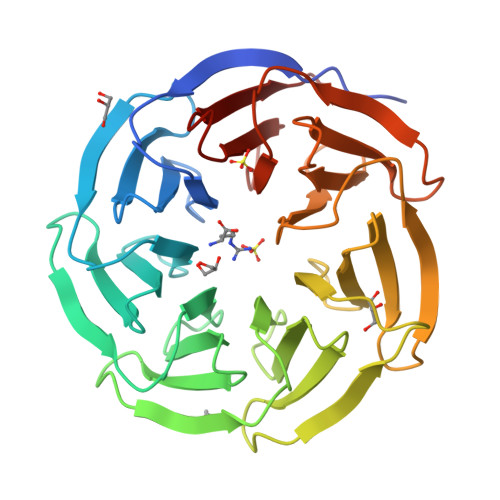
A Binary Arginine Methylation Switch on Histone H3 Arginine 2 Regulates Its Interaction with WDR5.
Lorton, B.M., Harijan, R.K., Burgos, E.S., Bonanno, J.B., Almo, S.C., Shechter, D.(2020) Biochemistry 59: 3696-3708
- PubMed: 32207970
- DOI: https://doi.org/10.1021/acs.biochem.0c00035
- Primary Citation of Related Structures:
6OFZ, 6OI0, 6OI1, 6OI2, 6OI3 - PubMed Abstract:
Histone H3 arginine 2 (H3R2) is post-translationally modified in three different states by "writers" of the protein arginine methyltransferase (PRMT) family. H3R2 methylarginine isoforms include PRMT5-catalyzed monomethylation (me1) and symmetric dimethylation (me2s) and PRMT6-catalyzed me1 and asymmetric dimethylation (me2a). WD-40 repeat-containing protein 5 (WDR5) is an epigenetic "reader" protein that interacts with H3R2. Previous studies suggested that H3R2me2s specified a high-affinity interaction with WDR5. However, our prior biological data prompted the hypothesis that WDR5 may also interact with H3R2me1. Here, using highly accurate quantitative binding analysis combined with high-resolution crystal structures of WDR5 in complex with unmodified (me0) and me1/me2s l-arginine amino acids and in complex with the H3R2me1 peptide, we provide a rigorous biochemical study and address long-standing discrepancies of this important biological interaction. Despite modest structural differences at the binding interface, our study supports an interaction model regulated by a binary arginine methylation switch: H3R2me2a prevents interaction with WDR5, whereas H3R2me0, -me1, and -me2s are equally permissive.
Organizational Affiliation:
Department of Biochemistry, Albert Einstein College of Medicine, Bronx, New York 10461, United States.