Functional and structural characterization of an ECF-type ABC transporter for vitamin B12.
Santos, J.A., Rempel, S., Mous, S.T., Pereira, C.T., Ter Beek, J., de Gier, J.W., Guskov, A., Slotboom, D.J.(2018) Elife 7
- PubMed: 29809140
- DOI: https://doi.org/10.7554/eLife.35828
- Primary Citation of Related Structures:
6FNP - PubMed Abstract:
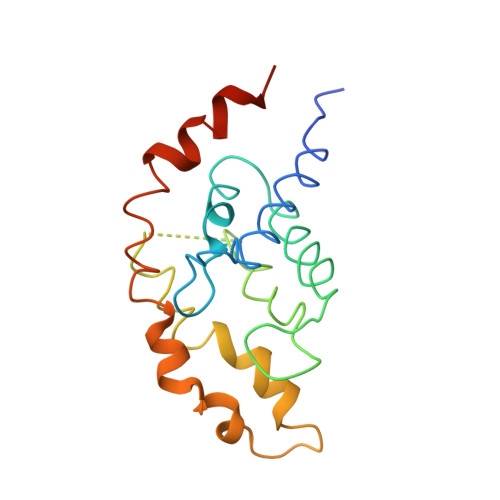
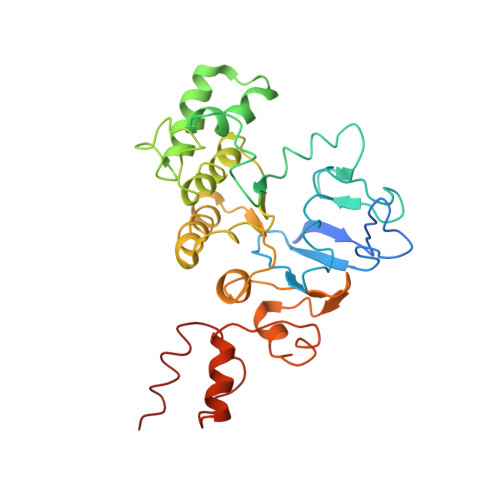
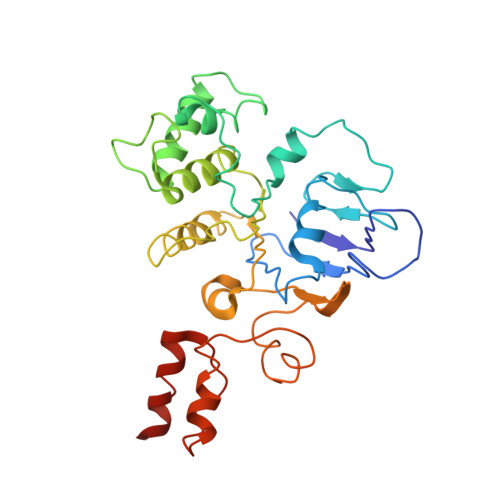
Vitamin B12 (cobalamin) is the most complex B-type vitamin and is synthetized exclusively in a limited number of prokaryotes. Its biologically active variants contain rare organometallic bonds, which are used by enzymes in a variety of central metabolic pathways such as L-methionine synthesis and ribonucleotide reduction. Although its biosynthesis and role as co-factor are well understood, knowledge about uptake of cobalamin by prokaryotic auxotrophs is scarce. Here, we characterize a cobalamin-specific ECF-type ABC transporter from Lactobacillus delbrueckii , ECF-CbrT, and demonstrate that it mediates the specific, ATP-dependent uptake of cobalamin. We solved the crystal structure of ECF-CbrT in an apo conformation to 3.4 Å resolution. Comparison with the ECF transporter for folate (ECF-FolT2) from the same organism, reveals how the identical ECF module adjusts to interact with the different substrate binding proteins FolT2 and CbrT. ECF-CbrT is unrelated to the well-characterized B12 transporter BtuCDF, but their biochemical features indicate functional convergence.
Organizational Affiliation:
Groningen Biomolecular and Biotechnology Institute (GBB), University of Groningen, Groningen, The Netherlands.