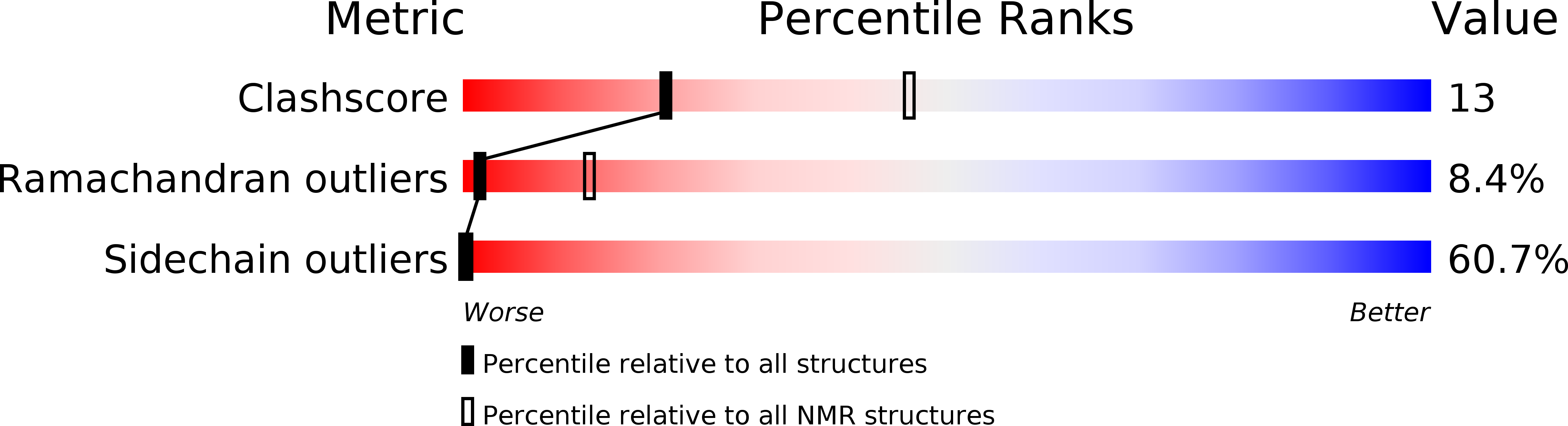Conformational studies of the N-terminal lipid-associating domain of human apolipoprotein C-I by CD and 1H NMR spectroscopy.
Rozek, A., Buchko, G.W., Kanda, P., Cushley, R.J.(1997) Protein Sci 6: 1858-1868
- PubMed: 9300485
- DOI: https://doi.org/10.1002/pro.5560060906
- Primary Citation of Related Structures:
1OPP - PubMed Abstract:
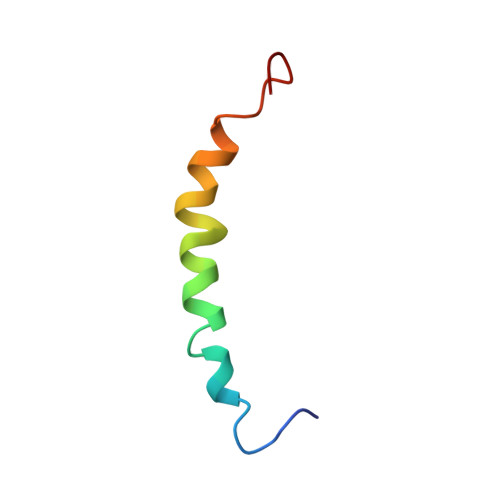
A peptide comprising the N-terminal 38 residues of human apolipoprotein C-I (apoC-I(1-38)) was synthesized using solid-phase methods and its solution conformation studied by CD and 1H NMR spectroscopy. The CD data indicate that apoC-I(1-38) has a similar helical content (55%) in the presence of saturating amounts of SDS or egg yolk lysophosphatidylcholine. A structural ensemble of SDS-bound apoC-I(1-38) was calculated from 464 NOE-based distance restraints using distance geometry methods. ApoC-I(1-38) adopts a helical structure between residues V4 and K30 and an extended C-terminus from Q31 when associated with SDS. The region K12-G15 undergoes slow conformational exchange as indicated by above-average amide resonance linewidths, large temperature coefficients, and fast exchange (< 2 h) of backbone amide protons with deuterium. The mobility of K12-G15 is reflected in the poorly defined dihedral angles of K12 and E13 in the calculated ensemble of structures. The average structure of apoC-I(1-38) is curved toward its hydrophobic face with bends of 125 degrees, centered at K12/E13, and 150 degrees, centered at K21. This curvature appears to be driven by the interaction of two hydrophobic clusters, one formed by residues L8, L11, F14, and L18, and the other by L25, I26, and I29, with the amphiphile SDS. Based on our present structural definition of apoC-I(1-38) and the previously obtained structure of the fragment apoC-I(35-53), we propose the secondary structure of intact apolipoprotein C-I.
Organizational Affiliation:
Department of Chemistry, Simon Fraser University, Burnaby, British Columbia, Canada.