Molecular determinants for Rous sarcoma virus intasome assemblies involved in retroviral integration.
Bera, S., Shi, K., Aihara, H., Grandgenett, D.P., Pandey, K.K.(2023) J Biol Chem 299: 104730-104730
- PubMed: 37084813
- DOI: https://doi.org/10.1016/j.jbc.2023.104730
- Primary Citation of Related Structures:
8E14 - PubMed Abstract:
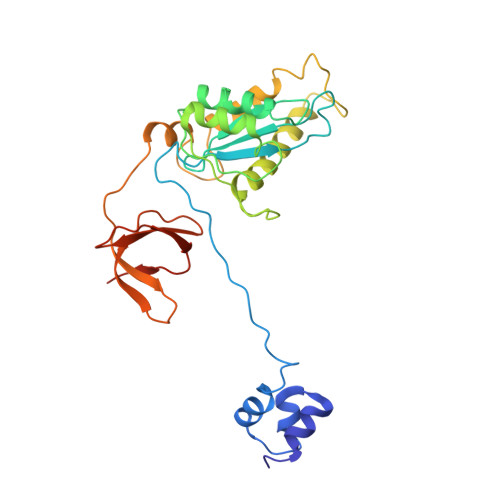
Integration of retroviral DNA into the host genome involves the formation of integrase (IN)-DNA complexes termed intasomes. Further characterization of these complexes is needed to understand their assembly process. Here, we report the single-particle cryo-EM structure of the Rous sarcoma virus (RSV) strand transfer complex (STC) intasome produced with IN and a preassembled viral/target DNA substrate at 3.36 Å resolution. The conserved intasome core region consisting of IN subunits contributing active sites interacting with viral/target DNA has a resolution of 3 Å. Our structure demonstrated the flexibility of the distal IN subunits relative to the IN subunits in the conserved intasome core, similar to results previously shown with the RSV octameric cleaved synaptic complex intasome produced with IN and viral DNA only. An extensive analysis of higher resolution STC structure helped in the identification of nucleoprotein interactions important for intasome assembly. Using structure-function studies, we determined the mechanisms of several IN-DNA interactions critical for assembly of both RSV intasomes. We determined the role of IN residues R244, Y246, and S124 in cleaved synaptic complex and STC intasome assemblies and their catalytic activities, demonstrating differential effects. Taken together, these studies advance our understanding of different RSV intasome structures and molecular determinants involved in their assembly.
Organizational Affiliation:
Department of Molecular Microbiology and Immunology, School of Medicine, Saint Louis University, St Louis, Missouri, USA.