A pH-dependent anti-CD47 antibody that selectively targets solid tumors and improves therapeutic efficacy and safety.
Li, Y., Liu, J., Chen, W., Wang, W., Yang, F., Liu, X., Sheng, Y., Du, K., He, M., Lyu, X., Li, H., Zhao, L., Wei, Z., Wang, F., Zheng, S., Sui, J.(2023) J Hematol Oncol 16: 2-2
- PubMed: 36650558
- DOI: https://doi.org/10.1186/s13045-023-01399-4
- Primary Citation of Related Structures:
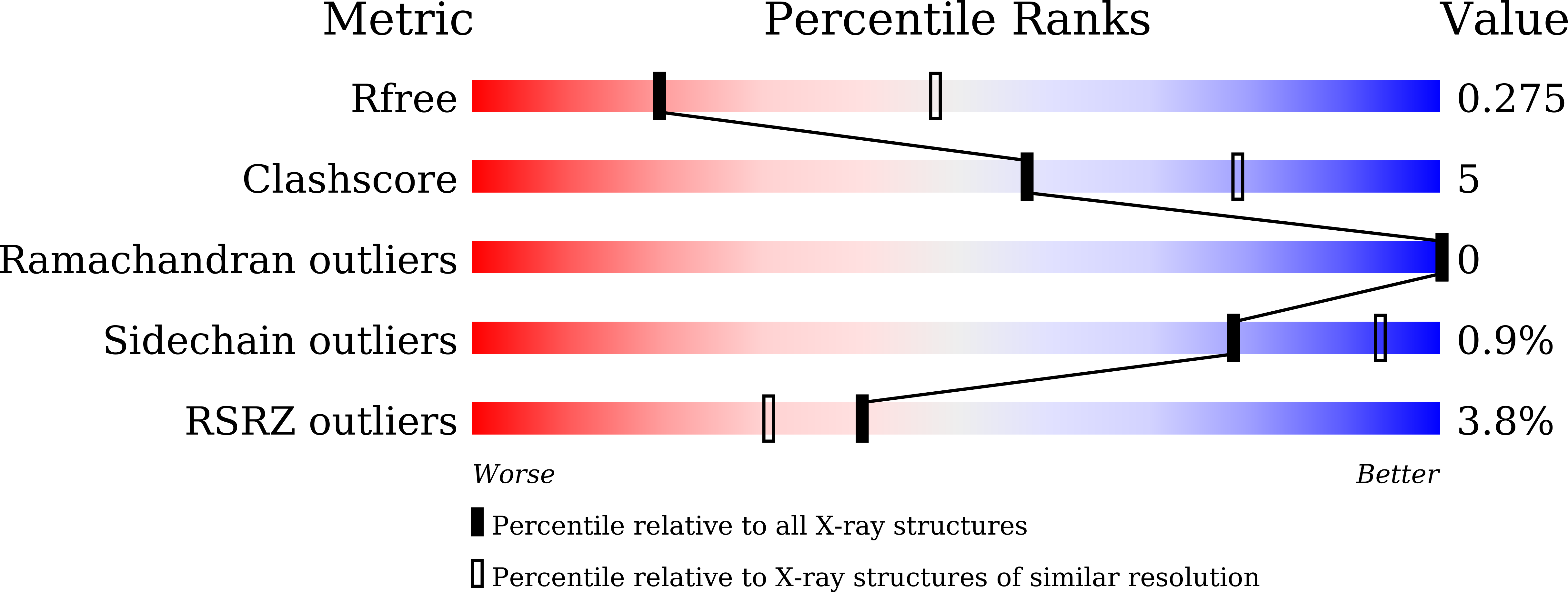
7WN8 - PubMed Abstract:
The antiphagocytic molecule CD47 is overexpressed in a wide variety of cancer cells, and antibodies targeting CD47 for cancer therapies are currently under intensive investigation. However, owing to the ubiquitous expression of CD47 on healthy cells, anti-CD47 therapies often achieve only weak therapeutic benefits and can induce severe side effects. Here, we report the generation of a pH-dependent anti-CD47 antibody (BC31M4) which selectively binds to tumors under the acidic solid tumor microenvironment. BC31M4 was generated using antibody phage display and a pH-dependent selection strategy. The pH-dependent binding and blocking activities of BC31M4 were verified using in vitro assays, and the structural basis of the pH-dependent binding property was characterized. BC31M4's antitumor effect was confirmed by both phagocytosis assays and studies in xenograft models. The tumor selectivity, mechanism of action, PK properties, side effects, and therapeutic efficacy were further evaluated in humanized (hCD47 and its receptor hSIRPα) immunocompetent syngeneic mouse models. The crystal structure reveals that two histidines locate within the CDRs of the light chain directly contribute to the pH-dependent binding of BC31M4. BC31M4 promotes macrophage phagocytosis of tumor cells more potently at acidic-pH than at physiological-pH. Our hCD47/hSIRPα humanized syngeneic mouse model results demonstrated that BC31M4 selectively accumulates in tumors but not in normal tissues. BC31M4 causes minimal side effects and exhibits superior PK properties as compared to the other examined anti-CD47 antibodies. When combined with adoptive T cell transfer, BC31M4 efficiently promotes adaptive immune responses against tumors and also induces immune memory. Moreover, we show that BC31M4's antitumor effects rely on an Fc that mediates strong effector functions. Our study illustrates that the development of a tumor-selective, pH-dependent anti-CD47 antibody safely confers strong therapeutic effects against solid tumors, thus providing a promising therapeutic strategy to overcome the challenges of anti-CD47 therapy.
Organizational Affiliation:
Peking University-Tsinghua University-National Institute of Biological Sciences (PTN) Joint Graduate Program, School of Life Sciences, Peking University, Beijing, China.