De novo mapping of alpha-helix recognition sites on protein surfaces using unbiased libraries.
Li, K., Tokareva, O.S., Thomson, T.M., Wahl, S.C.T., Travaline, T.L., Ramirez, J.D., Choudary, S.K., Agarwal, S., Walkup 4th, W.G., Olsen, T.J., Brennan, M.J., Verdine, G.L., McGee, J.H.(2022) Proc Natl Acad Sci U S A 119: e2210435119-e2210435119
- PubMed: 36534810
- DOI: https://doi.org/10.1073/pnas.2210435119
- Primary Citation of Related Structures:
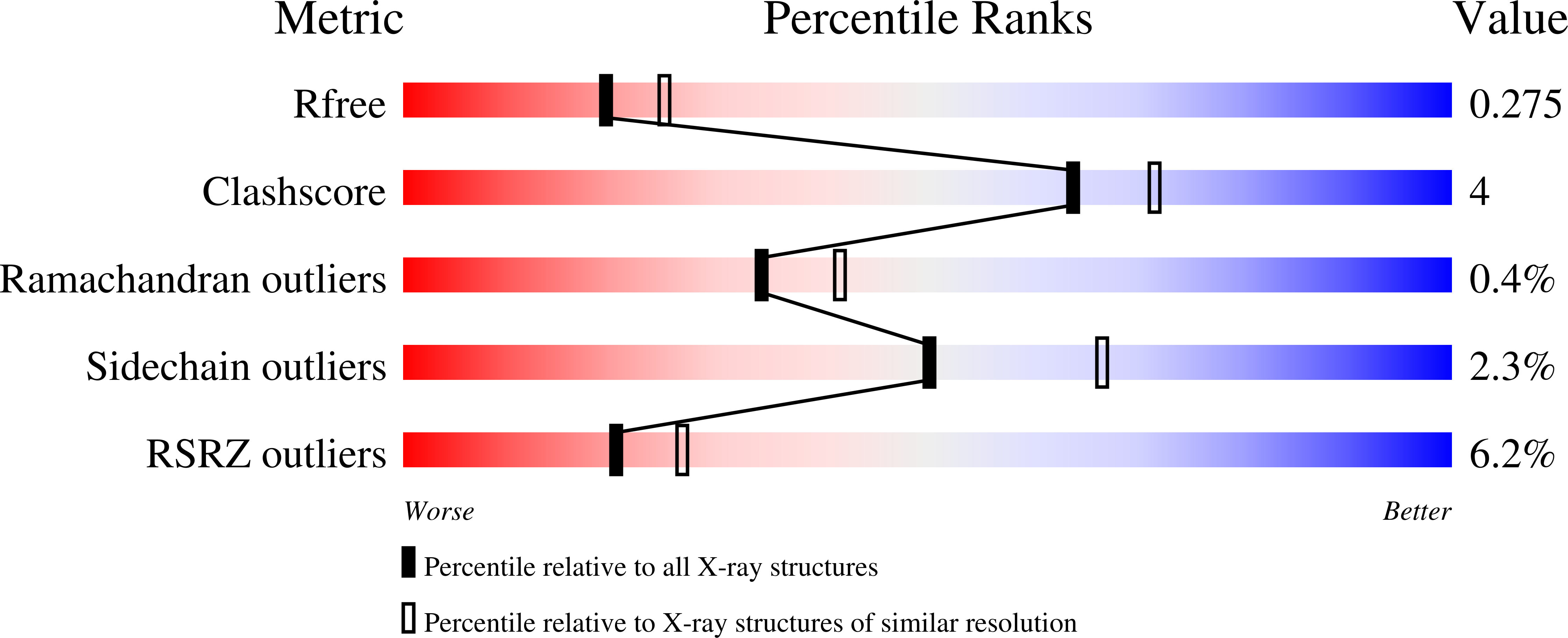
7UWI, 7UWO, 7UX5, 7UXI, 7UXJ, 7UXK, 7UXM, 7UXN, 7UXO, 7UXP, 7UXQ, 7UY2, 7UYJ, 7UYK - PubMed Abstract:
The α-helix is one of the most common protein surface recognition motifs found in nature, and its unique amide-cloaking properties also enable α-helical polypeptide motifs to exist in membranes. Together, these properties have inspired the development of α-helically constrained (Helicon) therapeutics that can enter cells and bind targets that have been considered "undruggable", such as protein-protein interactions. To date, no general method for discovering α-helical binders to proteins has been reported, limiting Helicon drug discovery to only those proteins with previously characterized α-helix recognition sites, and restricting the starting chemical matter to those known α-helical binders. Here, we report a general and rapid screening method to empirically map the α-helix binding sites on a broad range of target proteins in parallel using large, unbiased Helicon phage display libraries and next-generation sequencing. We apply this method to screen six structurally diverse protein domains, only one of which had been previously reported to bind isolated α-helical peptides, discovering 20 families that collectively comprise several hundred individual Helicons. Analysis of 14 X-ray cocrystal structures reveals at least nine distinct α-helix recognition sites across these six proteins, and biochemical and biophysical studies show that these Helicons can block protein-protein interactions, inhibit enzymatic activity, induce conformational rearrangements, and cause protein dimerization. We anticipate that this method will prove broadly useful for the study of protein recognition and for the development of both biochemical tools and therapeutics for traditionally challenging protein targets.
Organizational Affiliation:
FOG Pharmaceuticals Inc., Cambridge, MA 02140.