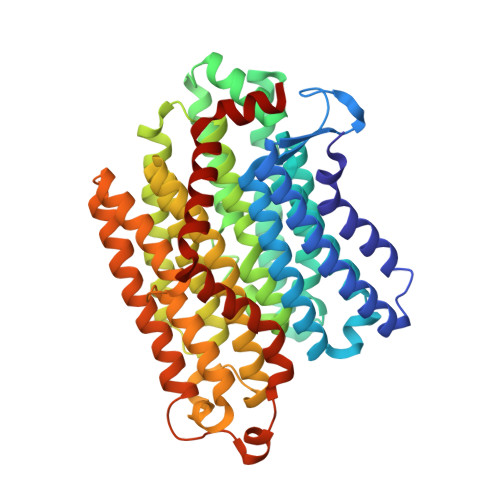
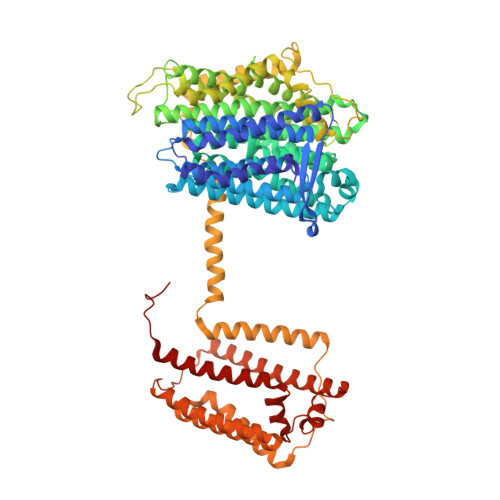
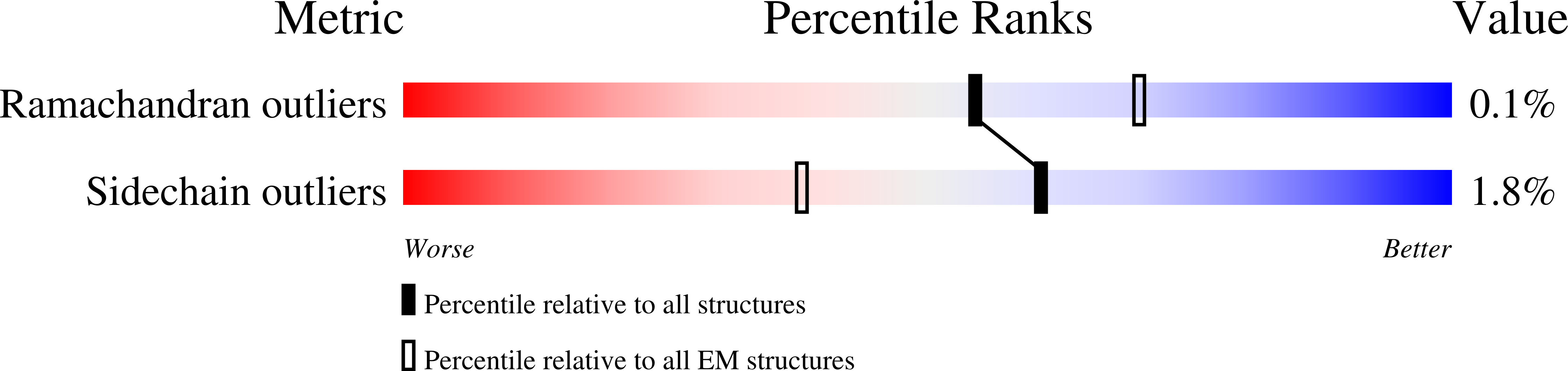Ion transfer mechanisms in Mrp-type antiporters from high resolution cryoEM and molecular dynamics simulations.
Lee, Y., Haapanen, O., Altmeyer, A., Kuhlbrandt, W., Sharma, V., Zickermann, V.(2022) Nat Commun 13: 6091-6091
- PubMed: 36241630
- DOI: https://doi.org/10.1038/s41467-022-33640-y
- Primary Citation of Related Structures:
7QRU - PubMed Abstract:
Multiple resistance and pH adaptation (Mrp) cation/proton antiporters are essential for growth of a variety of halophilic and alkaliphilic bacteria under stress conditions. Mrp-type antiporters are closely related to the membrane domain of respiratory complex I. We determined the structure of the Mrp antiporter from Bacillus pseudofirmus by electron cryo-microscopy at 2.2 Å resolution. The structure resolves more than 99% of the sidechains of the seven membrane subunits MrpA to MrpG plus 360 water molecules, including ~70 in putative ion translocation pathways. Molecular dynamics simulations based on the high-resolution structure revealed details of the antiport mechanism. We find that switching the position of a histidine residue between three hydrated pathways in the MrpA subunit is critical for proton transfer that drives gated trans-membrane sodium translocation. Several lines of evidence indicate that the same histidine-switch mechanism operates in respiratory complex I.
Organizational Affiliation:
Department of Structural Biology, Max Planck Institute of Biophysics, 60438, Frankfurt am Main, Germany.