A PROSS-designed extensively mutated estrogen receptor alpha variant displays enhanced thermal stability while retaining native allosteric regulation and structure.
Kriegel, M., Wiederanders, H.J., Alkhashrom, S., Eichler, J., Muller, Y.A.(2021) Sci Rep 11: 10509-10509
- PubMed: 34006920
- DOI: https://doi.org/10.1038/s41598-021-89785-1
- Primary Citation of Related Structures:
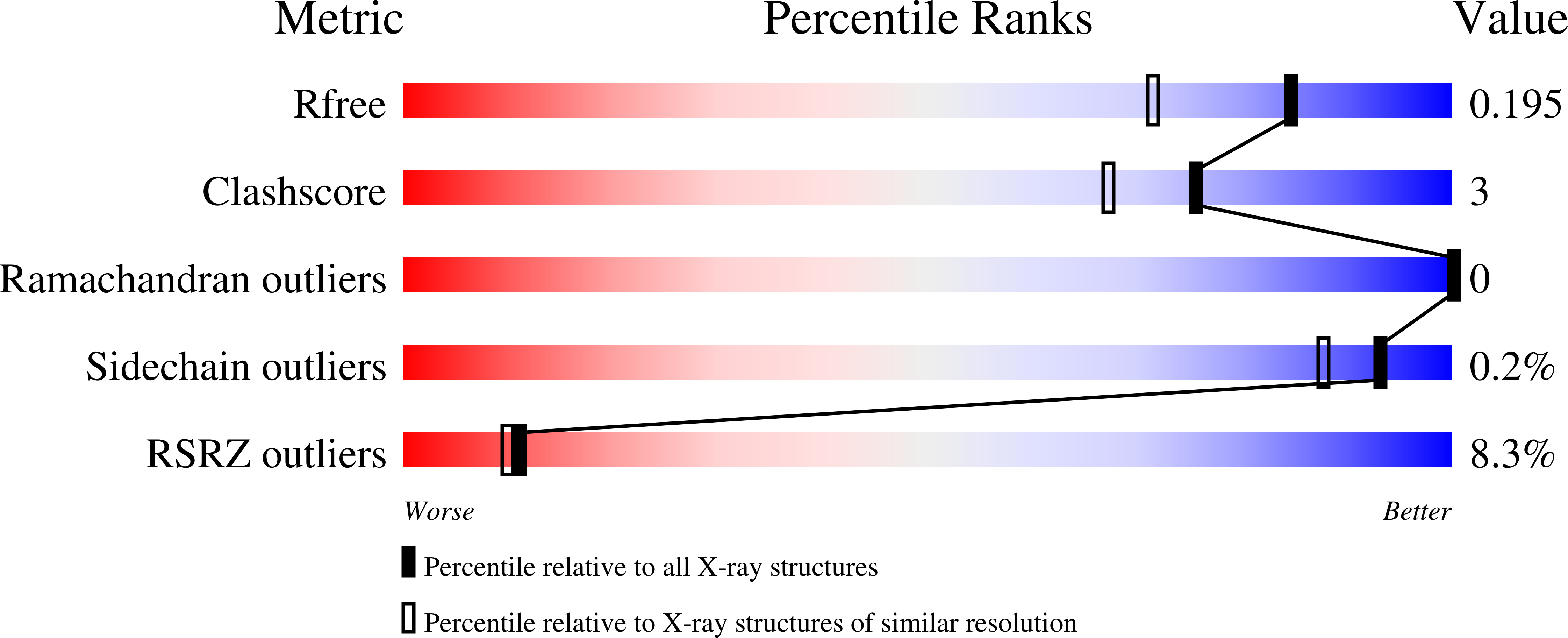
7NDO, 7NEL, 7NFB - PubMed Abstract:
Protein stability limitations often hamper the exploration of proteins as drug targets. Here, we show that the application of PROSS server algorithms to the ligand-binding domain of human estrogen receptor alpha (hERα) enabled the development of variant ER PRS* that comprises 24 amino acid substitutions and exhibits multiple improved characteristics. The protein displays enhanced production rates in E. coli, crystallizes readily and its thermal stability is increased significantly by 23 °C. hERα is a nuclear receptor (NR) family member. In NRs, protein function is allosterically regulated by its interplay with small molecule effectors and the interaction with coregulatory proteins. The in-depth characterization of ER PRS* shows that these cooperative effects are fully preserved despite that 10% of all residues were substituted . Crystal structures reveal several salient features, i.e. the introduction of a tyrosine corner in a helix-loop-helix segment and the formation of a novel surface salt bridge network possibly explaining the enhanced thermal stability. ER PRS* shows that prior successes in computational approaches for stabilizing proteins can be extended to proteins with complex allosteric regulatory behaviors as present in NRs. Since NRs including hERα are implicated in multiple diseases, our ER PRS* variant shows significant promise for facilitating the development of novel hERα modulators.
Organizational Affiliation:
Division of Biotechnology, Department of Biology, Friedrich-Alexander-Universität Erlangen-Nürnberg (FAU), Erlangen, Germany.