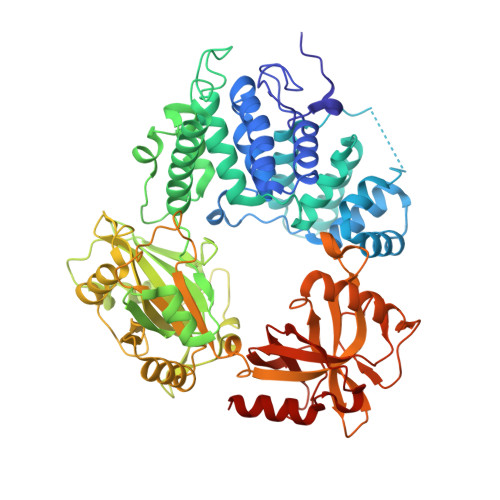
High-fidelity DNA ligation enforces accurate Okazaki fragment maturation during DNA replication.
Williams, J.S., Tumbale, P.P., Arana, M.E., Rana, J.A., Williams, R.S., Kunkel, T.A.(2021) Nat Commun 12: 482-482
- PubMed: 33473124
- DOI: https://doi.org/10.1038/s41467-020-20800-1
- Primary Citation of Related Structures:
7KR3, 7KR4 - PubMed Abstract:
DNA ligase 1 (LIG1, Cdc9 in yeast) finalizes eukaryotic nuclear DNA replication by sealing Okazaki fragments using DNA end-joining reactions that strongly discriminate against incorrectly paired DNA substrates. Whether intrinsic ligation fidelity contributes to the accuracy of replication of the nuclear genome is unknown. Here, we show that an engineered low-fidelity LIG1 Cdc9 variant confers a novel mutator phenotype in yeast typified by the accumulation of single base insertion mutations in homonucleotide runs. The rate at which these additions are generated increases upon concomitant inactivation of DNA mismatch repair, or by inactivation of the Fen1 Rad27 Okazaki fragment maturation (OFM) nuclease. Biochemical and structural data establish that LIG1 Cdc9 normally avoids erroneous ligation of DNA polymerase slippage products, and this protection is compromised by mutation of a LIG1 Cdc9 high-fidelity metal binding site. Collectively, our data indicate that high-fidelity DNA ligation is required to prevent insertion mutations, and that this may be particularly critical following strand displacement synthesis during the completion of OFM.
Organizational Affiliation:
Genome Integrity and Structural Biology Laboratory, National Institute of Environmental Health Sciences, US National Institutes of Health, Department of Health and Human Services, 111 TW Alexander Drive, Research Triangle Park, NC, 27709, USA.