Insights into the mismatch discrimination mechanism of Y-family DNA polymerase Dpo4.
Jung, H., Lee, S.(2021) Biochem J 478: 1769-1781
- PubMed: 33881499
- DOI: https://doi.org/10.1042/BCJ20210162
- Primary Citation of Related Structures:
6VKP - PubMed Abstract:
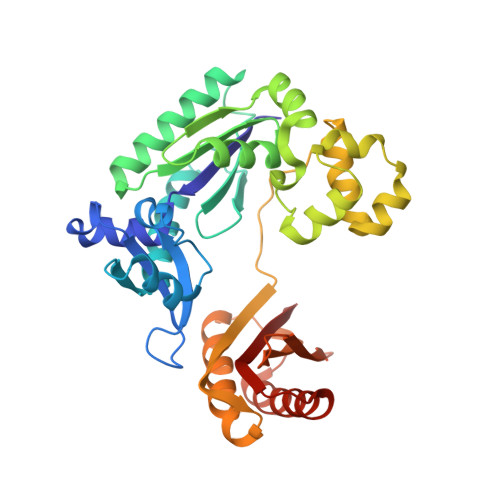
Nucleobases within DNA are attacked by reactive oxygen species to produce 7,8-dihydro-8-oxoguanine (oxoG) and 7,8-dihydro-8-oxoadenine (oxoA) as major oxidative lesions. The high mutagenicity of oxoG is attributed to the lesion's ability to adopt syn-oxoG:anti-dA with Watson-Crick-like geometry. Recent studies have revealed that Sulfolobus solfataricus P2 DNA polymerase IV (Dpo4) inserts nucleotide opposite oxoA in an error-prone manner and accommodates syn-oxoA:anti-dGTP with Watson-Crick-like geometry, highlighting a promutagenic nature of oxoA. To gain further insights into the bypass of oxoA by Dpo4, we have conducted kinetic and structural studies of Dpo4 extending oxoA:dT and oxoA:dG by incorporating dATP opposite templating dT. The extension past oxoA:dG was ∼5-fold less efficient than that past oxoA:dT. Structural studies revealed that Dpo4 accommodated dT:dATP base pair past anti-oxoA:dT with little structural distortion. In the Dpo4-oxoA:dG extension structure, oxoA was in an anti conformation and did not form hydrogen bonds with the primer terminus base. Unexpectedely, the dG opposite oxoA exited the primer terminus site and resided in an extrahelical site, where it engaged in minor groove contacts to the two immediate upstream bases. The extrahelical dG conformation appears to be induced by the stabilization of anti-oxoA conformation via bifurcated hydrogen bonds with Arg332. This unprecedented structure suggests that Dpo4 may use Arg332 to sense 8-oxopurines at the primer terminus site and slow the extension from the mismatch by promoting anti conformation of 8-oxopurines.
Organizational Affiliation:
The Division of Chemical Biology and Medicinal Chemistry, College of Pharmacy, The University of Texas at Austin, Austin, Texas 78712, U.S.A.