Discovery of novel liver X receptor inverse agonists as lipogenesis inhibitors.
Chen, Z., Chen, H., Zhang, Z., Ding, P., Yan, X., Li, Y., Zhang, S., Gu, Q., Zhou, H., Xu, J.(2020) Eur J Med Chem 206: 112793-112793
- PubMed: 32961480
- DOI: https://doi.org/10.1016/j.ejmech.2020.112793
- Primary Citation of Related Structures:
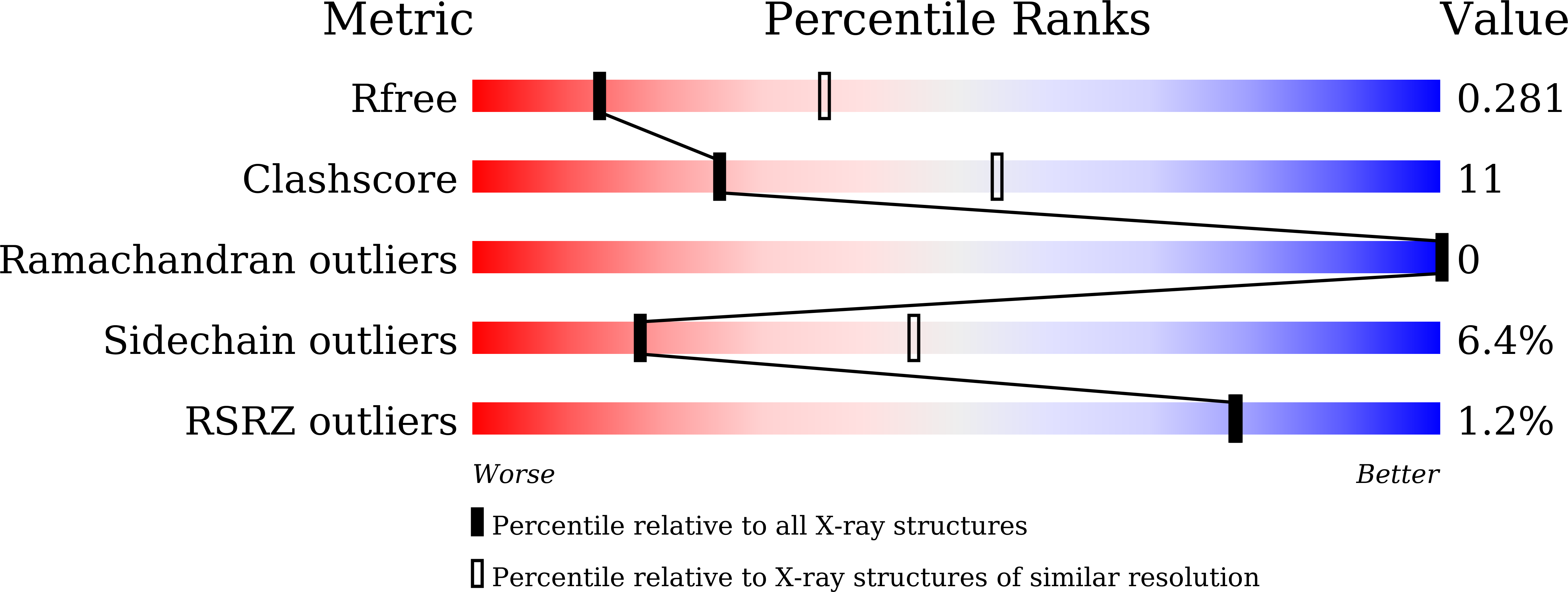
6K9M - PubMed Abstract:
Based on the co-crystal structures of LXRβ and its agonists (spiro [pyrrolidine-3,3'-oxindole] derivatives) discovered by us previously, we designed and synthesized a compound library to explore the agonistic activities. The library was screened with luciferase reporter assays, interestingly, it resulted in the discovery of 10 LXR inverse agonists besides 5 LXR agonists. To clarify the mechanism of the actions, we conducted molecular dynamics (MD) simulations on the LXR and inverse agonists complexes, and revealed that H3, H11 and H12 configurations are the key to turn on agonism or inverse agonism status for LXR. Binding tightly with H3, pushing H11 out and destabilizing H12 could form a bigger hydrophobic groove to accommodate NCOR1 to turn on LXR inverse agonism. The inverse agonist 10rr was further studied, and found as a lipogenesis inhibitor through down-regulating LXR target genes SREBP-1c, ACC, FAS and SCD-1, and demonstrated lipid-lowering effects in 3T3-L1 cells, HepG2 cells and mice with Triton WR-1339-induced hyperlipidemia. Therefore, we have proved that LXR inverse agonists can be promising agents for hyperlipidemia treatment.
Organizational Affiliation:
Research Center for Drug Discovery, School of Pharmaceutical Sciences, Sun Yat-Sen University, Guangzhou, 510006, China; Shenzhen Pingle Orthopaedic Hospital (Shenzhen Pingshan Traditional Chinese Medicine Hospital), Shenzhen, 518118, China.