A higher-order configuration of the heterodimeric DOT1L-AF10 coiled-coil domains potentiates their leukemogenenic activity.
Song, X., Yang, L., Wang, M., Gu, Y., Ye, B., Fan, Z., Xu, R.M., Yang, N.(2019) Proc Natl Acad Sci U S A 116: 19917-19923
- PubMed: 31527241
- DOI: https://doi.org/10.1073/pnas.1904672116
- Primary Citation of Related Structures:
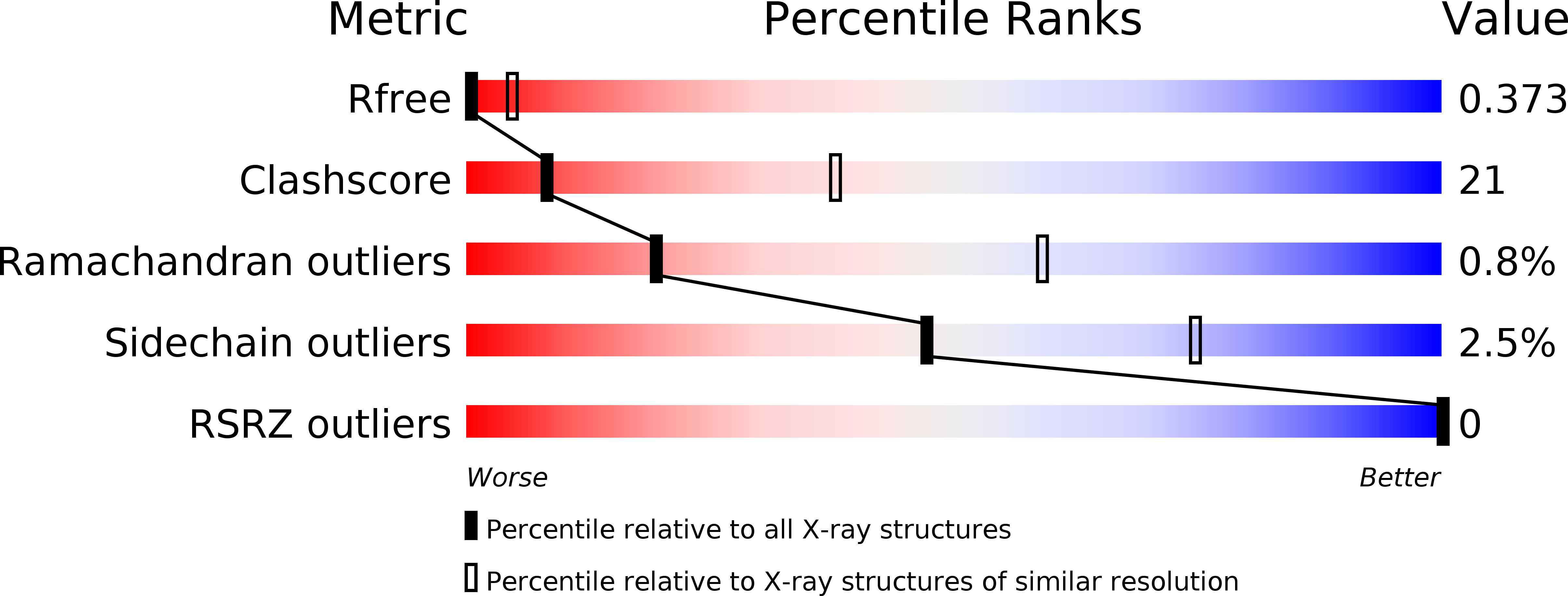
6JN2 - PubMed Abstract:
Chromosomal translocations of MLL1 (Mixed Lineage Leukemia 1) yield oncogenic chimeric proteins containing the N-terminal portion of MLL1 fused with distinct partners. The MLL1-AF10 fusion causes leukemia through recruiting the H3K79 histone methyltransferase DOT1L via AF10's octapeptide and leucine zipper (OM-LZ) motifs. Yet, the precise interaction sites in DOT1L, detailed interaction modes between AF10 and DOT1L, and the functional configuration of MLL1-AF10 in leukeomogenesis remain unknown. Through a combined approach of structural and functional analyses, we found that the LZ domain of AF10 interacts with the coiled-coil domains of DOT1L through a conserved binding mode and discovered that the C-terminal end of the LZ domain and the OM domain of AF10 mediate the formation of a DOT1L-AF10 octamer via tetramerization of the binary complex. We reveal that the oligomerization ability of the DOT1L-AF10 complex is essential for MLL1-AF10's leukemogenic function. These findings provide insights into the molecular basis of pathogenesis by MLL1 rearrangements.
Organizational Affiliation:
State Key Laboratory of Medicinal Chemical Biology, College of Pharmacy and Tianjin Key Laboratory of Molecular Drug Research, Nankai University, 300353 Tianjin, China.