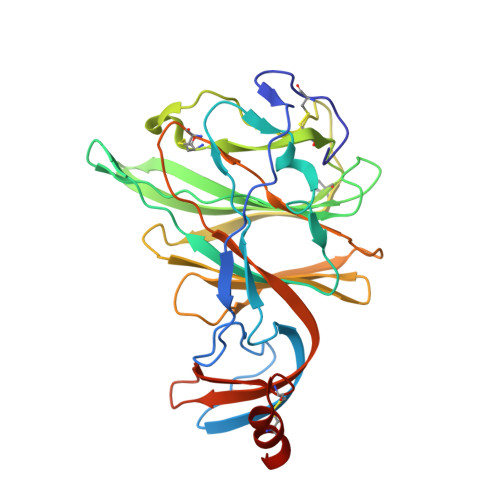
Crystal structure of the S1 subunit N-terminal domain from DcCoV UAE-HKU23 spike protein.
Cheng, Y., He, B., Yang, J., Ye, F., Lin, S., Yang, F., Chen, Z., Chen, Z., Cao, Y., Lu, G.(2019) Virology 535: 74-82
- PubMed: 31279241
- DOI: https://doi.org/10.1016/j.virol.2019.06.015
- Primary Citation of Related Structures:
6JHY - PubMed Abstract:
The DcCoV UAE-HKU23 coronavirus is a newly-found betacoronavirus (betaCoV) that can infect human cells. The viral spike protein plays pivotal roles in mediating receptor-recognition and membrane-fusion, and is therefore a key factor involved in viral pathogenesis and inter-species transmission. Here we reported the structural and functional characterization of the spike N-terminal domain (NTD) from DcCoV UAE-HKU23 (HKU23-NTD). Via mucin-binding assays, we showed that HKU23-NTD is able to bind sugars. We further solved the structure of HKU23-NTD, performed structure-guided mutagenesis and successfully located the potential sugar-binding pockets in the structure. Furthermore, via comparison of available betaCoV NTD structures, we demonstrated that betaCoV NTDs contain a conserved β-sandwich core, but exhibit variant folds in the peripheral elements located in the top-ceiling region and on the lateral side. While showing different compositions and structures, these peripheral elements are topologically equivalent β-sandwich-core insertions, highlighting a divergent evolution process for betaCoVs to form different lineages.
Organizational Affiliation:
West China Hospital Emergency Department (WCHED), State Key Laboratory of Biotherapy, West China Hospital, Sichuan University, and Collaborative Innovation Center of Biotherapy, Chengdu, Sichuan, 610041, China; Disaster Medicine Center, Sichuan University, Chengdu, Sichuan, 610041, China.