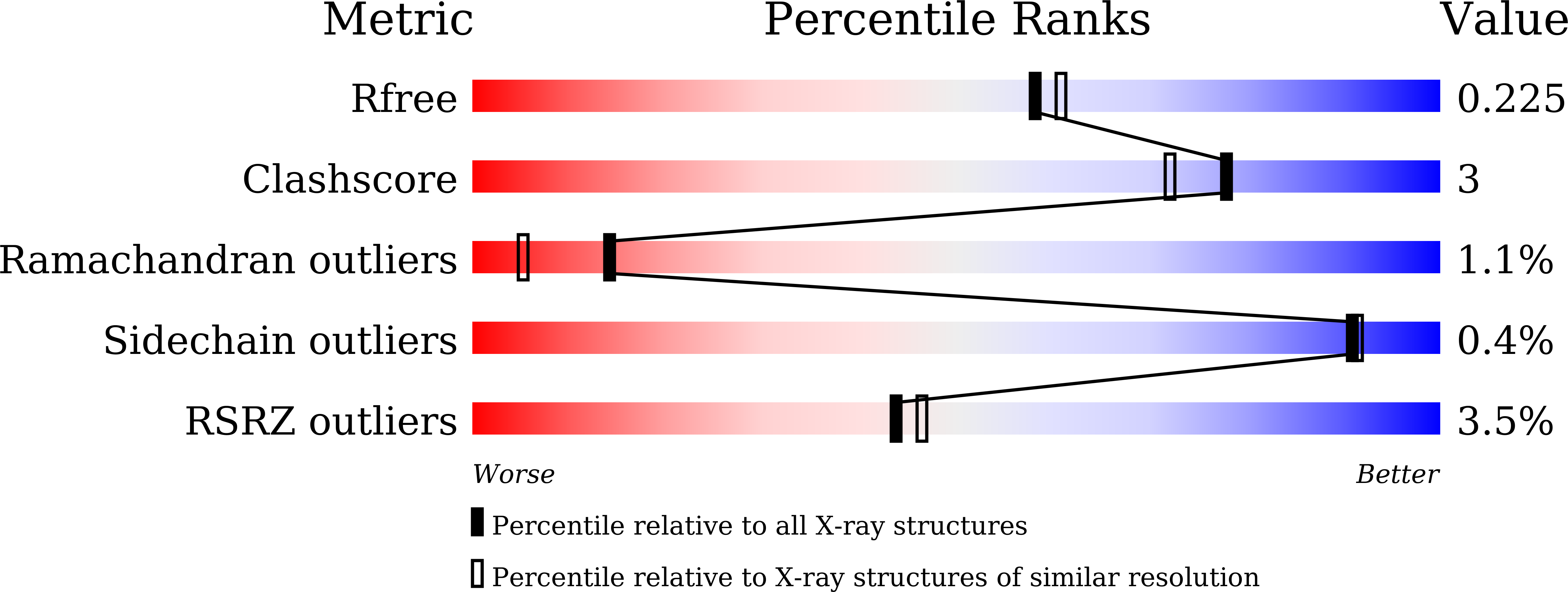
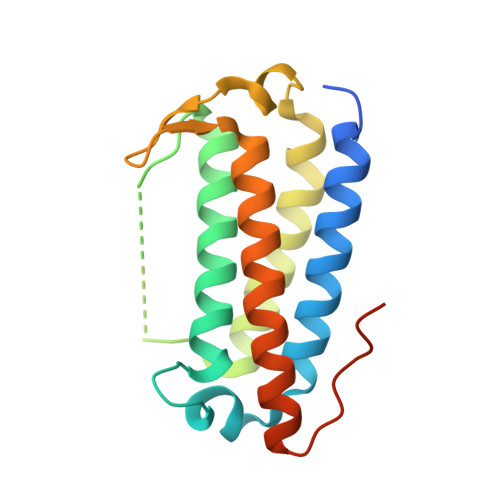
Crystal structure of the highly radiation-inducible DinB/YfiT superfamily protein DR0053 from Deinococcus radiodurans R1.
Zhang, J., Zhao, L., Seo, H.S., Jung, J.H., Choi, J.I., Kim, M.K., Lim, S.(2019) Biochem Biophys Res Commun 513: 354-359
- PubMed: 30961930
- DOI: https://doi.org/10.1016/j.bbrc.2019.03.209
- Primary Citation of Related Structures:
6IZ2 - PubMed Abstract:
Deinococcus radiodurans is an extremophilic bacterium well-known for its extraordinary resistance to ionizing radiation and other DNA damage- and oxidative stress-generating agents. In addition to its efficient DNA damage repair and oxidative stress resistance mechanisms, protein family expansions and stress-induced genes/proteins are also regarded as important components that add to the robustness of this bacterium. D. radiodurans encodes specific expansions of 13 DinB/YfiT homologs, which is a relatively large number when compared to those found in Gram-positive bacteria. In this study, we investigated the expression profiles of 13 dinB genes after γ-irradiation, mitomycin C and H 2 O 2 treatment. dr0053 had the highest expression levels after DNA-damage inducing γ-irradiation and MMC treatment, increasing ∼200-fold and ∼16-fold, respectively. We also determined the crystal structure of DR0053 at 2.07 Å resolution. DR0053 adopted a typical four-helix bundle structure that is characteristic of DinB/YfiT proteins. A putative metal binding site was occupied by zinc even though the highly conserved His triad of DinB/YfiT proteins was replaced by Glu-Asn-His.
Organizational Affiliation:
Biotechnology Research Division, Korea Atomic Energy Research Institute, Jeongeup, 56212, Republic of Korea; Department of Biotechnology and Bioengineering, Interdisciplinary Program for Bioenergy and Biomaterials, Chonnam National University, Gwangju, 61186, Republic of Korea.