The structure of DLP12 endolysin exhibits likely an active and inactive conformations.
Kesavan, B., Arockiasamy, A., Krishnaswamy, S.To be published.
Experimental Data Snapshot
wwPDB Validation 3D Report Full Report
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Lysozyme RrrD | 175 | Escherichia coli K-12 | Mutation(s): 0 Gene Names: rrrD, arrD, ybcS, b0555, JW0544 EC: 3.2.1.17 | 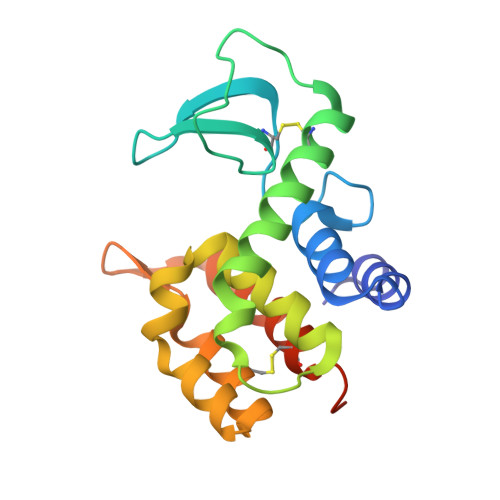 | |
UniProt | |||||
Find proteins for P78285 (Escherichia coli (strain K12)) Explore P78285 Go to UniProtKB: P78285 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P78285 | ||||
Sequence AnnotationsExpand | |||||
| |||||
| Ligands 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| ACT Query on ACT | E [auth A], F [auth B], G [auth C], J [auth D] | ACETATE ION C2 H3 O2 QTBSBXVTEAMEQO-UHFFFAOYSA-M |  | ||
| FMT Query on FMT | H [auth C], I [auth C] | FORMIC ACID C H2 O2 BDAGIHXWWSANSR-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 78.106 | α = 90 |
| b = 94.131 | β = 90 |
| c = 97.43 | γ = 90 |
| Software Name | Purpose |
|---|---|
| REFMAC | refinement |
| HKL-2000 | data reduction |
| HKL-2000 | data scaling |
| PHENIX | phasing |